|
Poster week! This week we finally submitted our posters to the daunting finality of the South Slough office printer. It was exciting to have a tangible product that summarized all of the hard work we did this summer. On Wednesday we presented out posters at the symposium, and I realized how fun it is to tell other people about your research. Presenting science is definitely an art form that I am far from mastering, but it’s exciting to weave your research into a story that is understandable to people from various scientific backgrounds. Our plan research-wise this week was to see if our echinoderm nerve cell antibody (1E11) would work on any other closely-related taxa, such as the nemertean pilidium larvae. Since we didn’t have any other larval cultures in the lab, we decided to go out and do some plankton tows, collect any plankton of interest, and fix and stain them this week. Unfortunately, we did a several tows on Monday and Tuesday and came up short. It appears that there’s an uncharacteristically low amount of plankton for this time of year, so alas, our last experiment did not come to fruition. One of many lackluster plankton tows Last Saturday, we all went over to Maya’s house to pick blackberries and bake blackberry pie. I think I’ll be dreaming about that blackberry pie for a long time. On Tuesday, the REUs all went out to dinner to EZ Thai (highly recommend!!!) to celebrate the end of the program together. On Friday, we headed over to Richard’s house for a final potluck. It was a wonderful evening full of delicious food and flying potatoes. I’ve learned so much during my time at OIMB this summer, and I have a greater appreciation for all of the hard work that goes into research. There are so many things you don’t see when you read a paper – now, when I read some scientific articles, I can’t help but be in awe of the sheer amount of detail that goes into them. Although I am excited to return to my home institution with all of the things that I’ve learned, I’ll absolutely miss OIMB and the amazing people I’ve met here this summer. I mentioned in my first blog post that I hoped to become a part of this close-knit community, and I definitely think I and all the other REUs achieved that. I’ll miss the beautiful Oregon coast scenery, the long walks to Bastendorf Beach, the trips to Bayside Coffee, and even the smell of shrimp early in the morning. And of course, I'll miss the research and the people. Thank you OIMB for the amazing summer. I hope to be back again some day!
0 Comments
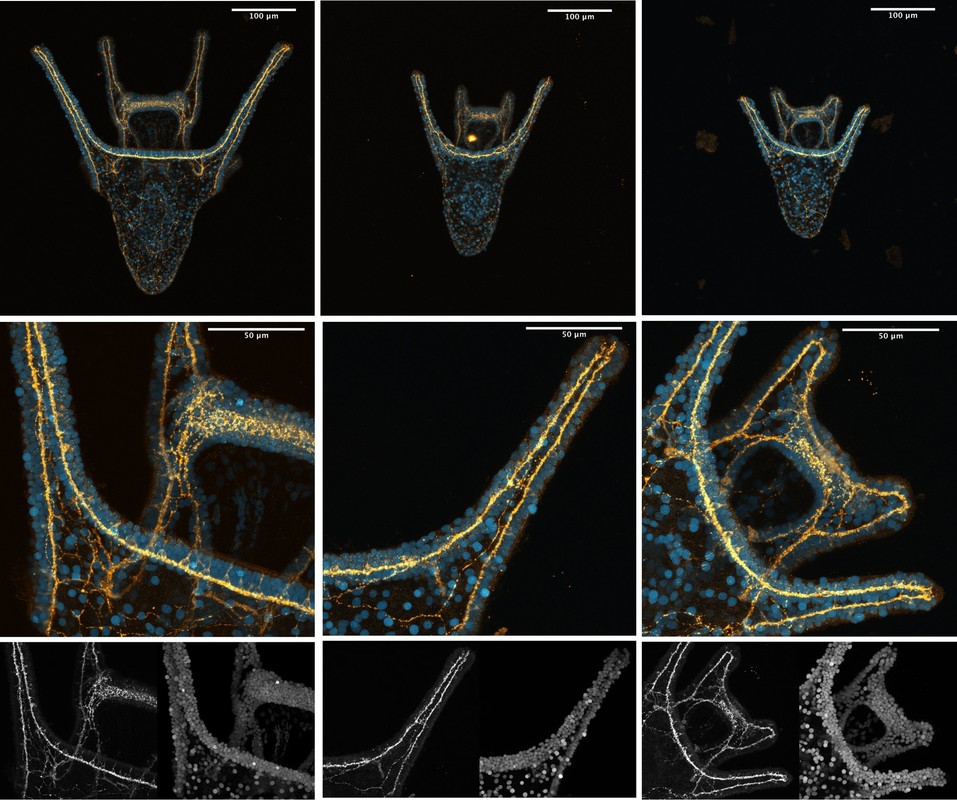
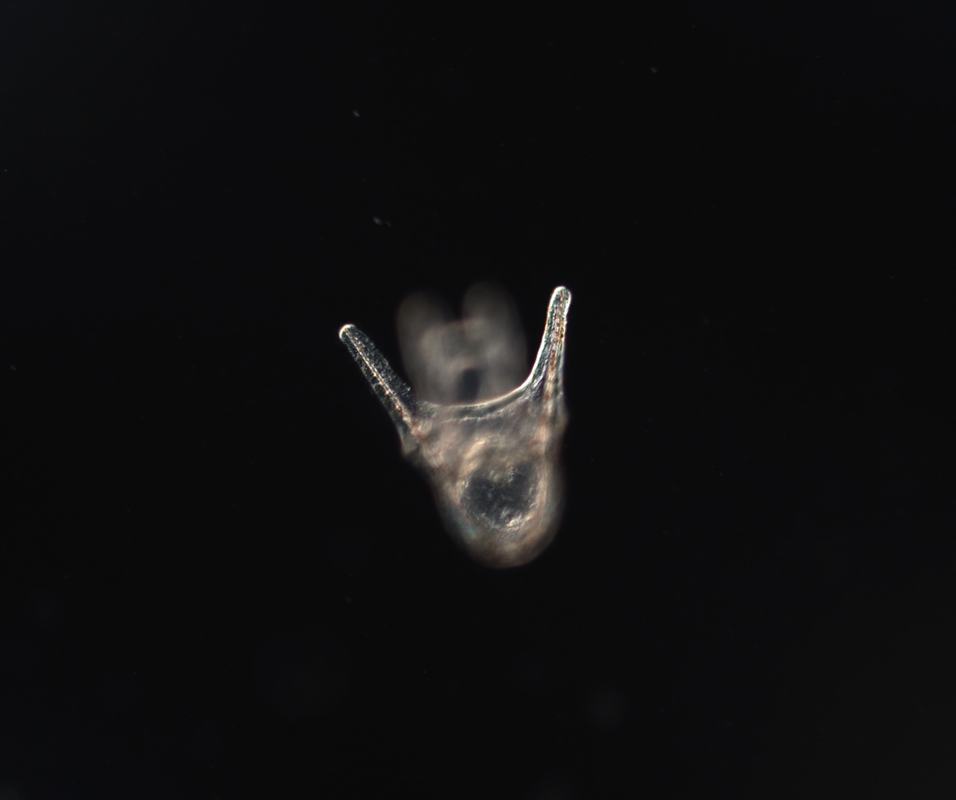
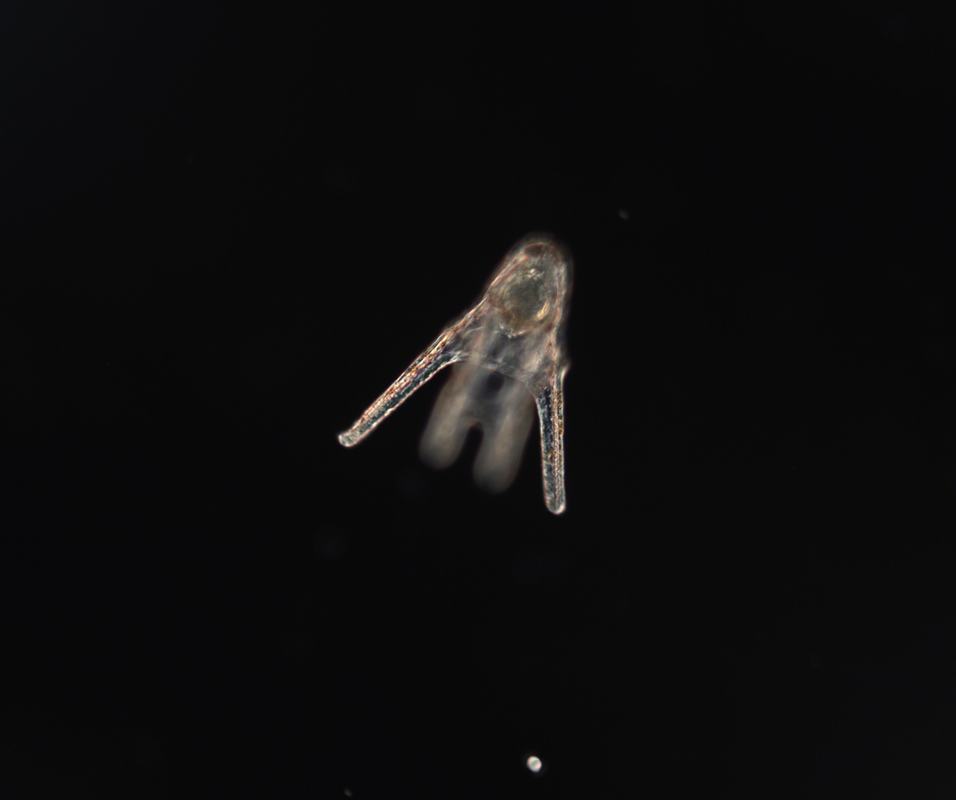
Hey there! Week 8 has been dedicated primarily to 1) creating our posters and 2) scrambling to collect any last-minute data we need in order to complete our posters. The last two weeks especially have involved a lot of late nights in lab and a few microscope-induced headaches, but it has been well worth it. After a “meh” staining earlier this week, we finally had success on Thursday! We got some beautiful pictures of larval nervous systems of 7-day old whole-, half-, and quarter-sized larvae and it was a great way to end several weeks of hard work. From left to right, these are images of 7-day old whole-, half-, and quarter-sized larvae stained with 1E11, a primary antibody that stains for synaptotagmin and thus allows us to visualize the larval nervous system. In yellow, you can see axons and nerve cell bodies, and in blue you can see the results of counterstaining with propidium iodide, which stains DNA and thus the nuclei of cells. We also managed to get some nice images of flow fields, which are essentially stacked images that display the movement of particles around a given larva. These images allow us to observe the velocity of particles moving around a larva and the load that larvae are moving. To do this, we start by placing a larva in a glass dish containing seawater with algae in it (in this case we used Isochrysis). We then chase around the larva with a teeny-tiny glass pipet that produces just enough suction by capillary action to stick onto the larva’s “butt.” This is a pretty difficult task, as any small movement can knock the larva off of the pipet tip. After many tries, I was finally able to get videos of whole-, half-, and quarter-sized larvae. These flow field images are informative of how well a given larva is able to produce localized currents, which are important in capturing food particles. Flow field images of whole, half- and quarter-sized larvae at 14 days old On Tuesday, the rough drafts of our posters were due. In our professional development session that day, we all projected our posters on the board and had a 10-minute rapid-fire critique session where everyone said what they did and didn’t like about each poster. This was definitely a bit intimidating, but it was very beneficial and it helped me see things that I wouldn’t have noticed myself.
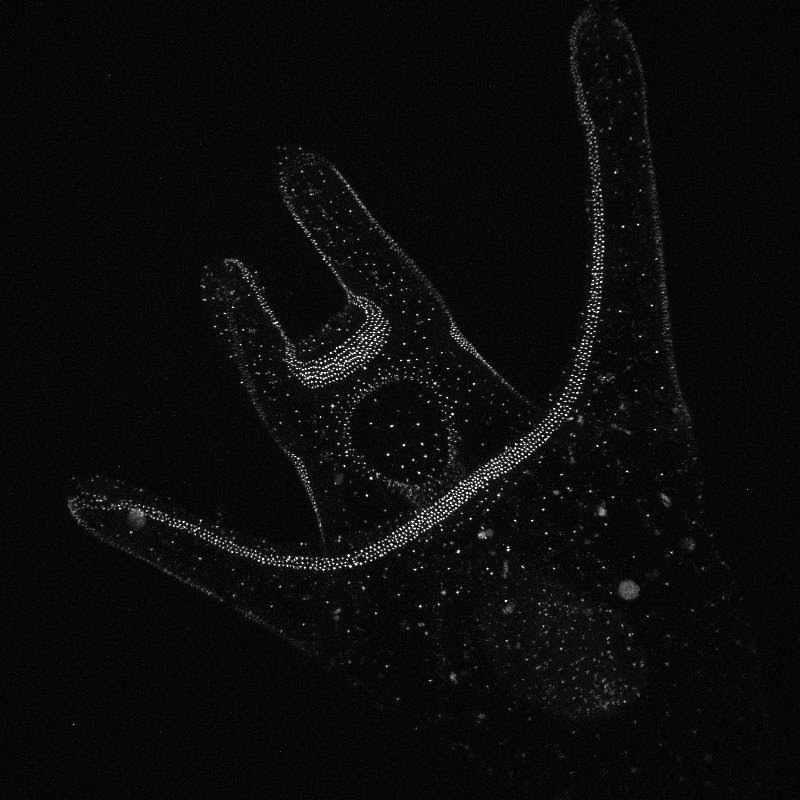
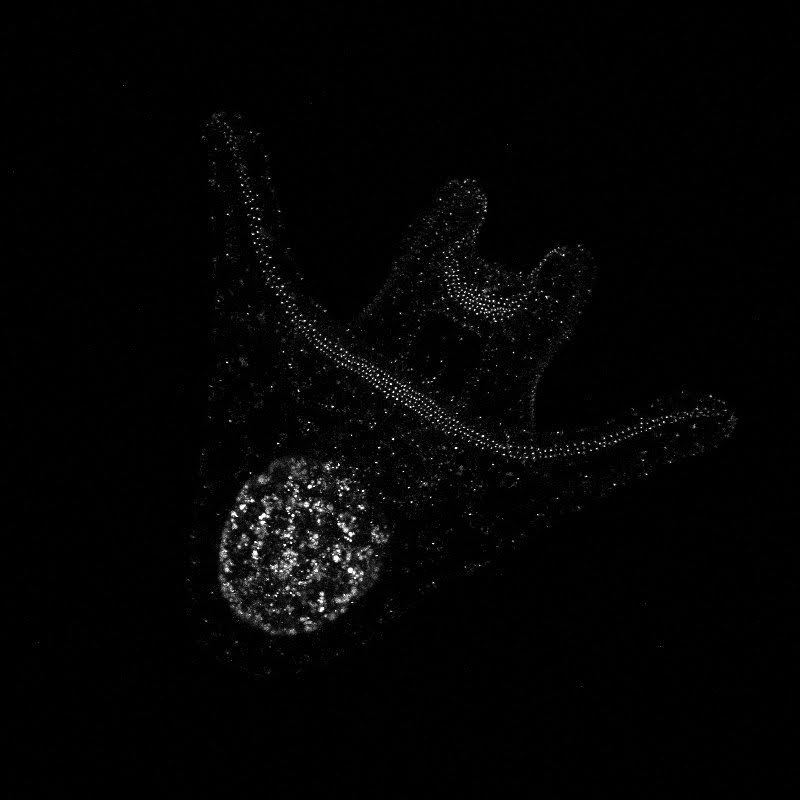
On Thursday, we got to attend the wonderful annual Invertebrate Ball! It was a busy week so, in all honesty, I most definitely slacked on my costume. However, Korrina was nice enough to lend me some “eel grass” (green ribbon) and a couple crab stickers before the event, so I was still able to walk the catwalk and participate in the festivities. The Invertebrate Ball was a great way to end a difficult week. Hello! It’s been a busy week here at OIMB – lots of late nights in the lab and cramming to collect all of our data before our posters are due. We’ve been documenting the development of our cultures of whole, half-, and quarter-sized sand dollar larvae, taking photos with the compound microscope every day. We also did antibody staining with some of the larvae that we had fixed in week 6, which should allow us to observe the nervous system under the confocal microscope and see if there are any observable differences between the wholes, halves, and quarters. The antibody staining was trickier than we expected. Since we didn’t have that many larvae to begin with (we can only separate so many blastomeres, and the war against bacteria continues) we didn’t want to sacrifice too many to the fate of fixation and staining. As a result, we only had a few larvae (anywhere from two to 14) floating around in the small vials in which they are preserved. To our surprise, when we went to do the antibody staining, we couldn’t even find half of our larvae! I don’t know where they went (our hypothesis is they traveled through a portal in the vial to another dimension), but spent a lot of time searching for these larvae that were M.I.A. In the end, we were able to save some, so here’s hoping that the staining process was successful and we’ll get some awesome pictures from the confocal! Centrin-labelled whole and quarter sand dollar larvae (left and right respectively) Note the ciliary band running along arms and around the mouth! In addition to antibody staining, we’ve been using high speed video to observe how effectively larvae are able to perform localized ciliary reversals and capture and retain food. This has involved a lot of sitting at the microscope, staring at a larva as it chows down on some yummy algae and trying to figure out which specific events are best to record. We just started two new cultures this past Wednesday and Thursday – we’ll be using the compound microscope to take measurements of arm length at different developmental stages throughout the next week, and we will also try to record more videos of feeding. We’ll be doing all of this on top of preparing our posters so that we can send them off to printing next Monday! A million things to do and not enough time, as usual.
On Thursday, we all presented our research to the public at the Charleston Marine Life Center. It was really fun to get to present our research to a broader audience and to brainstorm ways to explain things like blastomere separations to kids, teenagers, and adults. On Friday, we ventured to Eugene, where Dr. Kelly Sutherland gave us a tour of the science buildings at the University of Oregon main campus. We also visited the Museum of Natural and Cultural History and explored some of the shops downtown. It was a beautiful and sunny day in Eugene and a great way to end a moderately stressful week of research. It’s crunch time! We have less than three weeks before our poster presentations, which means we don’t have a lot more time left to collect data. Kostantina and I realized that we’ve probably bit off more than we can chew. We faced some challenges separating urchin and starfish blastomeres, so we’ve decided to try to simplify our projects a bit by focusing our efforts on sand dollars. We’ve had consistent results with the sand dollar blastomere separations and we’ve been documenting the development of whole, half-, and quarter-sized sand dollars throughout the summer. This week, we fixed some specimens to prepare them for antibody staining, and we also started using the high-speed camera to record feeding in sand dollar larvae. For the high-speed video, we’ve been using a particularly large and jumpy strain of Rhodomonas to see how well whole and partial larvae are able to capture and retain escape-prone prey. Five day old sand dollar larvae! Last weekend, we ventured up to Newport to tour the Hatfield Marine Science Center and visit the Newport Aquarium. The 2.5 hour drive up north along the coast was scenic and beautiful. It was really interesting to see Hatfield’s campus and to meet some of the REU’s and PI’s from their program. On Tuesday night, a few of us attended one of the concerts of the Oregon Coast Music Festival. I’m a huge music nerd, so it was so amazing to get to attend a concert while here. They performed one of my all-time favorite pieces, and the acoustics in the high school auditorium were actually pretty incredible. Also on Tuesday, we had a professional development session where we got some great advice from Richard, Svetlana, and OIMB grad students about applying to grad school. That was probably my favorite professional development session yet, and I feel really grateful for all of the valuable advice I’ve received this summer about pursuing a career in marine biology. Some of the grad students mentioned that when you do research your work ends up being on your mind almost all of the time. I’m definitely feeling that a bit right now… Everywhere I go, I’m thinking of my little larvae and about what other experiments I could possibly do. There are so many questions to explore and I am always wishing there was more time – I guess that’s what grad school is for!
We are officially halfway through the program!!! Kostantina and I have become more independent in lab, which means preparing cultures of half- and quarter-sized larvae on our own. This also means that there is a lot more trial and error and a short life expectancy for many unlucky embryos :( Some blastomere separations go smoothly, only to have the culture decimated by some evil bacteria a couple days later. Some seem to be going smoothly and then at the very end things don’t work out. Some don’t go smoothly at all in the first place. But that’s research I guess! It can be frustrating at times, especially given that time is limited, but when things do work it’s really rewarding. Looking at a happy and healthy sand dollar larva Last week I promised I’d go more into the details of my project and what exactly I’ll be doing in lab, so here I am! In order to understand the impacts of scaling on the functionality of the ciliary band, I also want to take a look at the effects of scaling on the larval nervous system. The ciliary band is the center of the larval nervous system and nerve cells are innervated throughout the ciliary band. So, let’s say that when we “shrink” down these larvae into halves or quarters, larval structures don’t scale proportionately and the larva appears to prioritize certain structures – like the ciliary band that is oh-so essential for feeding and swimming – over others. If this is the case, then other parts of the larva will need to compensate. Since the ciliary band and nervous system are so closely associated, it is possible that nerve cells would end up as one of the ones compensating. The loss of these nerve cells, which appear to play an important role in sensing and detecting prey, could end up reducing the functionality of the ciliary band. On the other hand, if there is proportionate scaling, it’s possible that the density of ciliary band cells will be so reduced that there aren’t enough cilia to effectively detect and capture prey. In order to observe ciliary band density, we’ll be using a fluorescent tag (GFP-centrin) that labels basal bodies, which are organelles found at the base of ciliated cells. This involves microinjecting eggs, a process which essentially consists of injecting eggs with a very tiny needle filled with RNA encoding a given fluorescent marker. We can then use a confocal microscope to compare ciliary band density in whole, half-, and quarter-sized larvae. In short, the confocal microscope is a microscope that eliminates all out-of-focus light and creates a 3D image of a fluorescently labeled specimen. To visualize nerve cells, we’ll stain larvae with a neuron-specific antibody called 1E11 and observe again with the confocal. Ideally, I’d like to get images of the ciliary band and nervous system for whole, half-, and quarter-sized larvae of sea urchins, sand dollars, and starfish. This will mostly depend on our blastomere-separating and culturing skills, so here’s to hoping things go a little better in this coming week! If time allows it, I’d also like to observe larvae using high-speed video to see what the impacts of scaling are on their ability to capture escape-prone prey. Kostantina and George looking at a half-sized sand dollar larva using the confocal microscope
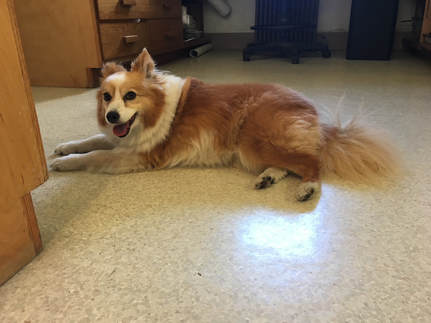
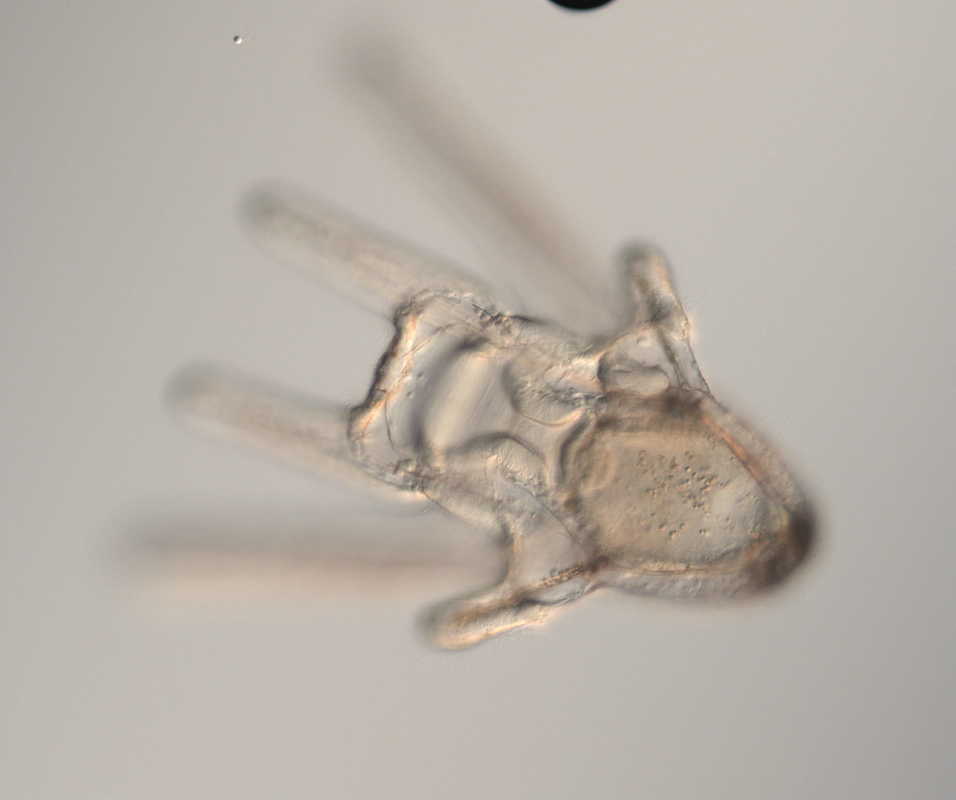
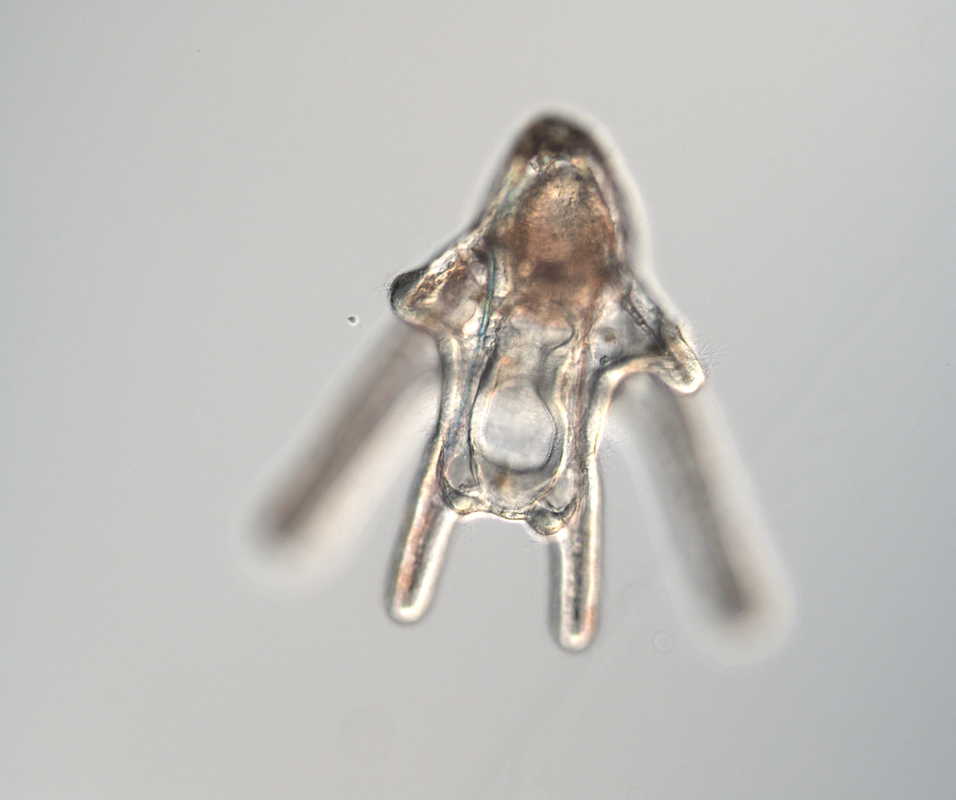
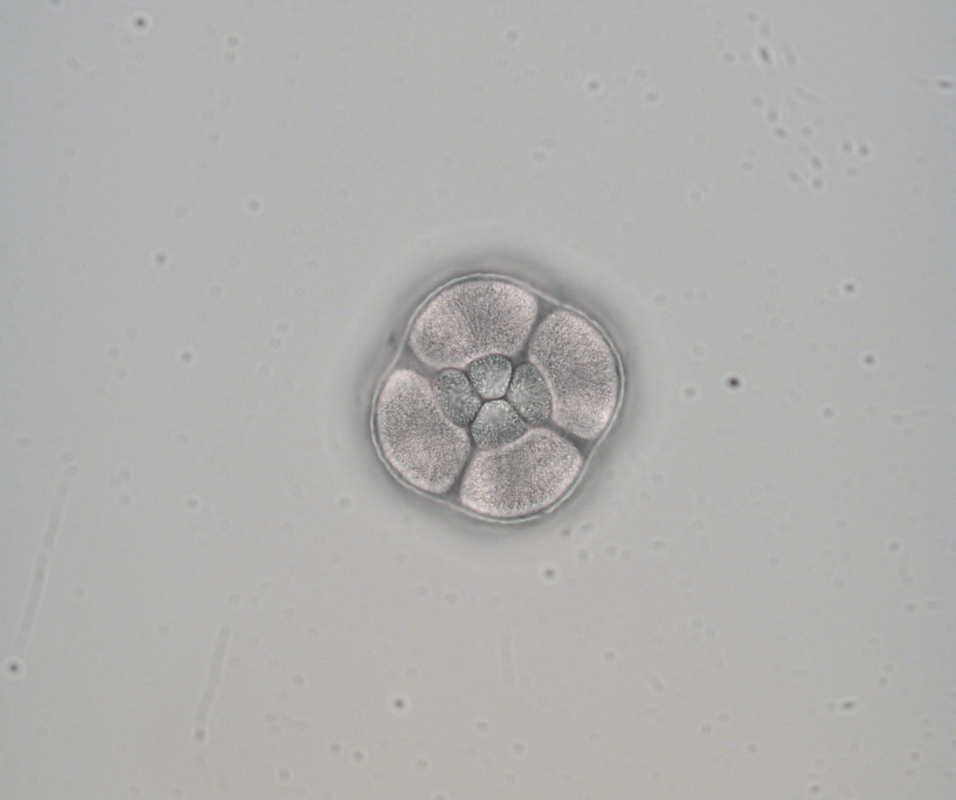
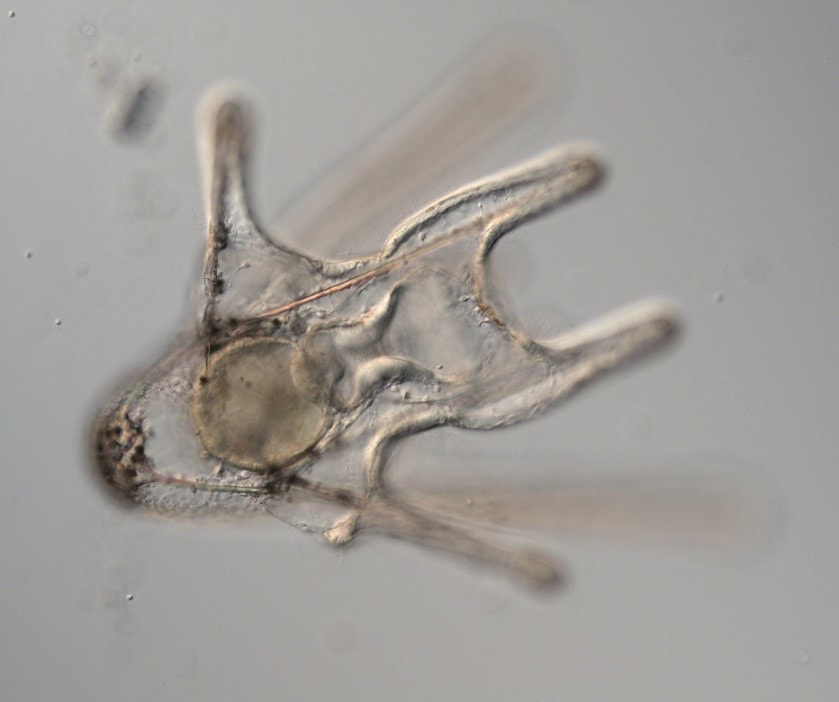
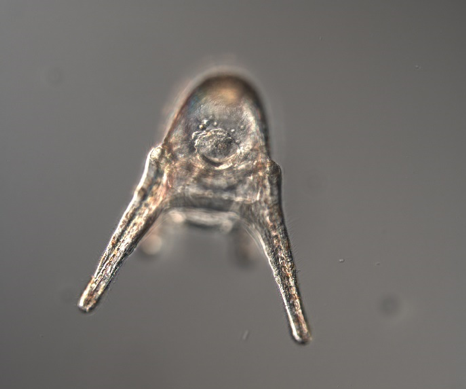
Hello, hello! This past week, our research proposals were due, meaning we all had to nail down our research questions and plan how we’ll work to answer them in the next six weeks. After a lot of thought, I’ve decided to investigate the feeding ability and morphological trade-offs of partial echinoderm larvae. When larvae are going about their lives in the ocean, they need to be able to collect food from their surroundings. Since algal cells are not particularly dense in ocean waters, it isn't efficient for larvae to just gulp up a bunch of water and filter it. Rather, they detect prey one-by-one and, ideally, capture and retain it. Central to this ability is the ciliary band – the principal feeding and swimming organ of a growing larva. In echinoderms, the ciliary band consists of a dense strip of ciliary cells that beat away from the mouth and, in response to detection of prey, produce local reversals of ciliary beat to redirect parcels of fluid toward the mouth (Strathmann 1975). Interestingly, purple sea urchins (Strongylocentrotus purpuratus) and bat stars (Patiria miniata), which have very small and large egg sizes respectively, are both able to produce functioning ciliary bands for feeding and swimming. I’m interested in understanding what happens to the ciliary band when a developing embryo loses a portion of this initial investment and must begin with a smaller starting point – will they still be able to produce a functioning ciliary band? If so, does this require other parts of the larva to compensate? Half- and quarter-sized embryos can develop into seemingly normal larvae. But what are the impacts of this manipulation on the ciliary band? One possibility is that the larva and all of its structures, including the ciliary band, scale down equally. Another possibility is that a partial larva would instead prioritize certain structures and cells over others, leading to an altered morphology. If the latter case is true, then the larva must compensate for these disproportionate structures, losing cells from certain areas to allow for a greater number of cells in others. By fluorescently tagging areas of interest, I hope to better understand the impacts of these embryological manipulations on the structure and function of the ciliary band in echinoderms. Next week, I’ll go more into the details of my project and how exactly I’ll be investigating these questions. In other news: Last weekend, a few interns and I made the trip down to California to do a little hiking and camping in the Redwoods! Our hike in Jedidiah Smith State Park was absolutely breathtaking. The first half of our hike consisted of a lot of silence interspersed with exclamations of amazement as we took in the nature around us. The way back went by a lot quicker, as we were (or at least I was) more preoccupied with the thought of s’mores awaiting us back at the campsite (did I mention I love s’mores?). On Tuesday, we discussed how to make scientific posters, and half of the REUs presented their research proposals. It's so great to hear about what everyone is doing and I'm so excited to see how everyone's projects turn out. DOG SPOTTING!!! Hello and welcome back! What I’ve learned this week is that there’s really no better way to start your morning than by shaking some urchins. Seriously, try it some time. I know it seems counterintuitive to get a good grip on a spiky ball in the palm of your hand and to shake it around forcefully, but it’s actually quite a magical trick. The most common way to spawn urchins is with an injection of potassium chloride (KCl) – this stimulates the gonad wall to contract and, if you’re lucky, lots of sperm or eggs will be released. A female can release millions of eggs during each spawning, and this is way more than we’re actually able to use in one experiment. When urchins are vigorously shaken, however, they only release a portion of their gametes, allowing us to use a given animal again for future spawning. Once they’re done, we put them back in the tank and let them relax for a while. As I gradually become more comfortable in the lab, time seems to go by more and more quickly. Last week, we started growing cultures of whole, half-, and quarter-sized sand dollar larvae in order to better understand how ciliary band and mouth proportions scale with changes in size. In order to create partial larvae, we must dissociate embryos at the 2- and 4-cell stages to form half- and quarter-sized larvae respectively. In addition, we injected one batch of embryos with GFP-centrin, allowing us to visualize ciliary band density using confocal microscopy. We have been like paparazzi or first-time moms with our little larvae, taking loads of pictures of them as they grow up. Left: Whole-sized sand dollar larva at 9 days old (20x magnification) Right: Half-sized sand dollar larva at 9 days old (20x magnification) In addition to creating partial sand dollar larvae, we began manipulations with purple sea urchin (Strongylocentrotus purpuratus) embryos. At the 16-cell stage, urchin embryos have 4 small cells called micromeres. These form on one side of the embryo and are responsible for development of the skeletal rods. Sounds pretty essential, right? Well, when these cells are removed, the embryo is not necessarily doomed – other cells around it can compensate and help to form skeletal rods. Alas, the larva survives… but not without some consequences. Micromere-less urchin embryos give rise to plutei with all the right parts but with altered proportions and undersized mouths. With these tiny-mouthed larvae, we can investigate the impacts of this manipulation on prey capture. The way we do this manipulation is, might I say, super cool. We use LASERS! We shoot embryos at the 16-cell stage with what is essentially a microscopic machine gun, taking out their micromeres and watching them explode under the microscope. It’s a great way to get out some frustration after a long day. Left: A wonderfully intact purple urchin embryo at the 16-cell stage Center: Two micromeres blasted, two more to go Right: A micromere-less purple urchin embryo Research alone has been a lot of fun, but we have had the opportunity to do a lot outside of the lab as well. Last weekend, we all went camping at Cape Arago along with students from the REU program at the main campus in Eugene. Some intense s’more making happened, and one REU may or may not have almost set his pants on fire with a flaming projectile marshmallow. On Sunday morning, we went tidepooling and I was blown away by the amazing biodiversity in the Oregon intertidal. We got to see urchin beds, nudibranchs, sea stars, anemones, sea cucumbers, crabs, and more. I have to say, tidepooling in the Pacific Northwest is really, really spectacular (sorry East Coast!). On the fourth of July, we attended the OIMB fourth of July picnic and participated in the annual egg toss. Juan and I made it to the top four thanks to a very, very tough egg that was willing to take a few falls. That night, we got to enjoy an amazing sunset and watched fireworks from the beach by the OIMB campus. I’ll say it every week, but time here is truly flying by and I am trying to cherish every moment! Thanks for reading!
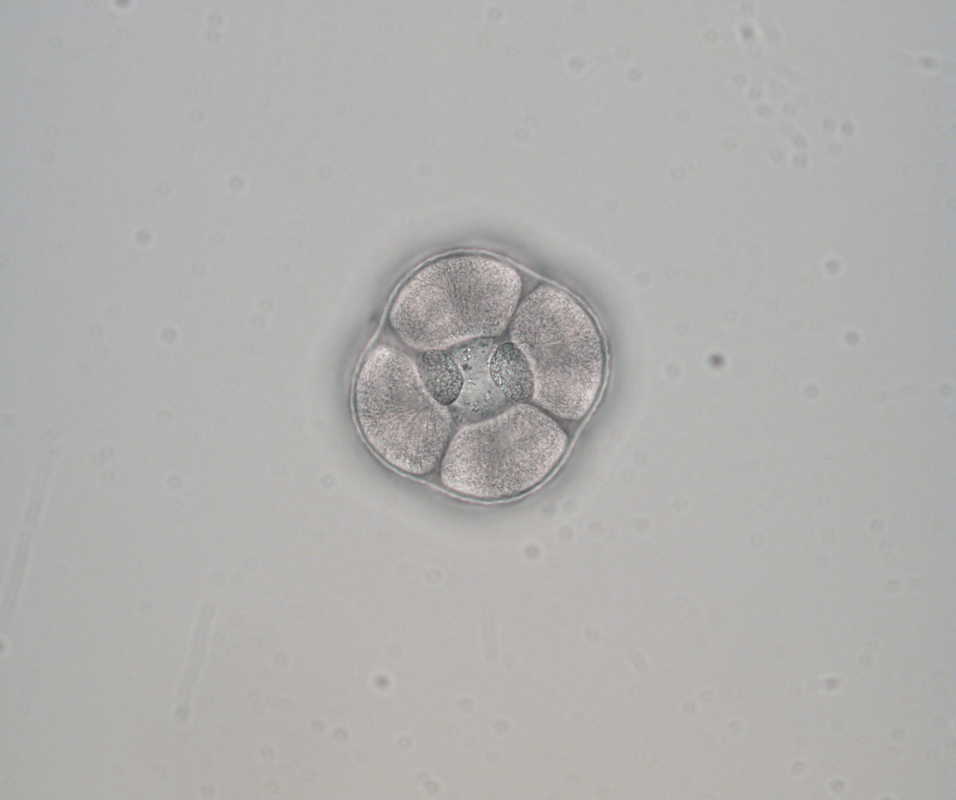
Week 2 is in the books! I’ve learned a lot in the lab in these past two weeks and have also enjoyed getting to know the other interns and hearing all about their projects. Last Saturday, we went out on the R/V Pluteus to do some dredging. Lots of fun, lots of waves, a little bit of motion sickness – but all in all an amazing day! We found a ton of cool stuff: basket stars, a variety of sea cucumbers, soft-body corals… On Sunday, Chris, Nancy, and I went on a short hike at Sunset Bay and Shore Acres State Park. It was absolutely beautiful and we couldn’t help but stop every few minutes to take pictures of the breathtaking views. We also had a professional development session this week in which we talked about research/science ethics and gave short presentations of our project proposals. Mia (Richard’s fabulous chihuahua) was a fantastic listener (see below). We spent most of last week learning the basic techniques we’ll need to do our experiments. This week, we started jumping into our project! Kostantina and I, with lots of help from our mentor George, will be investigating adaptations of ciliated larvae for prey capture. Before a sea urchin or star fish or sand dollar can be a sea urchin or star fish or sand dollar, it needs to grow up from a tiny embryo! Between the moment an egg is first fertilized by sperm and the juvenile phase, these invertebrates must go through a larval stage. During this time, these organisms look entirely different from their future adult forms. Looking at larvae under a microscope is like looking at clouds – some look like spaceships, some like giraffes, some like astronauts… Anyways, these larvae all have something in common – they use cilia (short, hairlike projections on the surface of certain cells) to swim and eat. These cilia can be found in the form of a ciliary band, which allows larvae to create currents to transport particles of food into their mouths. Invertebrate larvae, like humans, seem to have some foods they prefer and some that they’d rather avoid. One example of this is the nemertean pilidium larva, which appears to be incredibly efficient at capturing a type of algae called cryptomonads. Cryptomonads are a particularly good source of food: they are naked, nutrient-rich, easily-digestible cells. There is one caveat, though: cryptomonads have an escape response in which they essentially shoot out a microscopic thread that allows them to rapidly jump – a useful tool when trying to escape from a predator’s mouth. However, the pilidium larva is quite successful in eating these algae, and it does so by essentially not providing any indication to the prey that it’s being eaten until it’s too late! Before they know it, the cryptomonads are trapped in the pilidium’s large and deep mouth with no means of escape.
We are interested in understanding whether big, vestibular mouths help ciliated invertebrate larvae capture cryptomonad prey. We will be manipulating embryos of sand dollars, sea urchins, and star fish in order to create larvae of different sizes and size ratios and thus various mouth sizes. High-speed video will be used to determine whether it’s easier for prey to escape from small mouths, and confocal microscopy will be used to understand the impacts of our manipulations on larval proportions and the ciliary band. This week, we started a few cultures of half- and quarter-size sand dollar larvae, and we’ve been documenting their development using a compound microscope. In later weeks, I’ll get more into the nitty-gritty of the techniques we’re using! Thanks for reading! If you had told me a few years ago that I’d be studying marine biology, I probably wouldn’t have believed you. The ocean and all of the fascinating and unique life within it were not on my mind as I entered college with a plan to pursue a career in medicine. I began this plan by majoring in music while taking the courses necessary to apply to medical school: biology, general chemistry, organic chemistry, psychology… But when I finished my first year of college, I wasn’t satisfied – I wanted to understand, like really understand, the topics I had covered in those introductory biology courses. I wanted to know why and how things happened. I wanted to make more sense of the topics we had only just glossed over in that one year. I wanted to fall down the endless rabbit hole that is biology and to explore questions that didn’t necessarily have answers. I realized I wanted to embark on a journey of exploration and discovery in the sciences, continuously learning and trying to better understand the world around me.
I joined a marine organismal biology lab in my second year of college and discovered a field of study that I had never imagined myself in. I am so thankful for the unexpected opportunities that sometimes arise in life, because I couldn’t imagine myself doing anything else today. Whenever I’m not thinking about the ocean or science, I enjoy playing piano and bassoon, listening to podcasts, hiking, baking, eating ice cream (Moose Tracks especially), and hanging out with my dog and cats. This summer, I feel so lucky to be a part of the REU program here at OIMB and to have the opportunity to be immersed in research for an entire summer. I applied to the program because I wanted the opportunity to dedicate all of my time and energy to research and to spend an entire summer at a marine field station. As of right now, I am hoping to pursue a PhD in marine biology in the future, so the opportunity to work on my own project and to participate in professional development workshops is so valuable. This summer, I am working under the mentorship of George von Dassow, and I have already learned so much in just this first week. We spent the past few days learning some basic techniques that we’ll need for our projects, including microinjections and how to spawn, fertilize, and care for the various animals we will be working with. Alongside fellow REU Kostantina, I will likely be working on a project related to ciliated larvae and their adaptations for prey capture. I am looking forward to the rest of the summer and to seeing where my project takes me, and I hope this experience doesn’t go by too quickly! |
AuthorHello! My name is Ana and I am a rising senior studying biology and music at the College of the Holy Cross in Worcester, Massachusetts. This summer, I am working under the mentorship of George von Dassow. I am looking forward to seeing where my research takes me and to becoming a part of the OIMB community! Archives
August 2018
Categories |
Proudly powered by Weebly


































 RSS Feed
RSS Feed