|
Poster week! This week we finally submitted our posters to the daunting finality of the South Slough office printer. It was exciting to have a tangible product that summarized all of the hard work we did this summer. On Wednesday we presented out posters at the symposium, and I realized how fun it is to tell other people about your research. Presenting science is definitely an art form that I am far from mastering, but it’s exciting to weave your research into a story that is understandable to people from various scientific backgrounds. Our plan research-wise this week was to see if our echinoderm nerve cell antibody (1E11) would work on any other closely-related taxa, such as the nemertean pilidium larvae. Since we didn’t have any other larval cultures in the lab, we decided to go out and do some plankton tows, collect any plankton of interest, and fix and stain them this week. Unfortunately, we did a several tows on Monday and Tuesday and came up short. It appears that there’s an uncharacteristically low amount of plankton for this time of year, so alas, our last experiment did not come to fruition. One of many lackluster plankton tows Last Saturday, we all went over to Maya’s house to pick blackberries and bake blackberry pie. I think I’ll be dreaming about that blackberry pie for a long time. On Tuesday, the REUs all went out to dinner to EZ Thai (highly recommend!!!) to celebrate the end of the program together. On Friday, we headed over to Richard’s house for a final potluck. It was a wonderful evening full of delicious food and flying potatoes. I’ve learned so much during my time at OIMB this summer, and I have a greater appreciation for all of the hard work that goes into research. There are so many things you don’t see when you read a paper – now, when I read some scientific articles, I can’t help but be in awe of the sheer amount of detail that goes into them. Although I am excited to return to my home institution with all of the things that I’ve learned, I’ll absolutely miss OIMB and the amazing people I’ve met here this summer. I mentioned in my first blog post that I hoped to become a part of this close-knit community, and I definitely think I and all the other REUs achieved that. I’ll miss the beautiful Oregon coast scenery, the long walks to Bastendorf Beach, the trips to Bayside Coffee, and even the smell of shrimp early in the morning. And of course, I'll miss the research and the people. Thank you OIMB for the amazing summer. I hope to be back again some day!
0 Comments
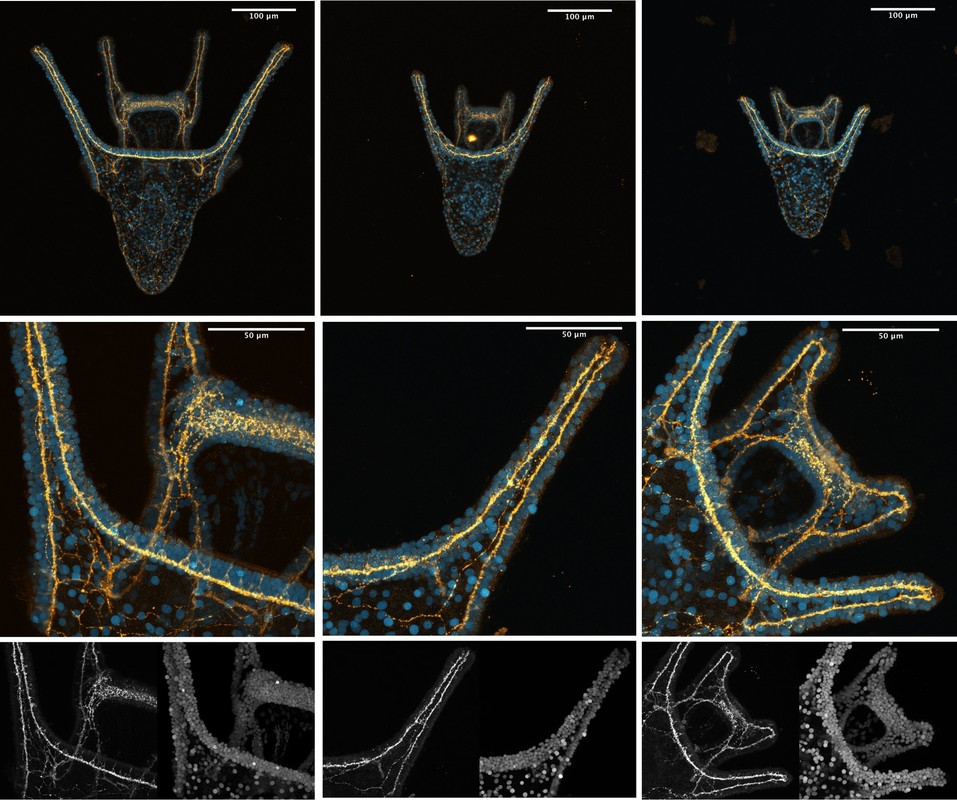
Hey there! Week 8 has been dedicated primarily to 1) creating our posters and 2) scrambling to collect any last-minute data we need in order to complete our posters. The last two weeks especially have involved a lot of late nights in lab and a few microscope-induced headaches, but it has been well worth it. After a “meh” staining earlier this week, we finally had success on Thursday! We got some beautiful pictures of larval nervous systems of 7-day old whole-, half-, and quarter-sized larvae and it was a great way to end several weeks of hard work. From left to right, these are images of 7-day old whole-, half-, and quarter-sized larvae stained with 1E11, a primary antibody that stains for synaptotagmin and thus allows us to visualize the larval nervous system. In yellow, you can see axons and nerve cell bodies, and in blue you can see the results of counterstaining with propidium iodide, which stains DNA and thus the nuclei of cells. We also managed to get some nice images of flow fields, which are essentially stacked images that display the movement of particles around a given larva. These images allow us to observe the velocity of particles moving around a larva and the load that larvae are moving. To do this, we start by placing a larva in a glass dish containing seawater with algae in it (in this case we used Isochrysis). We then chase around the larva with a teeny-tiny glass pipet that produces just enough suction by capillary action to stick onto the larva’s “butt.” This is a pretty difficult task, as any small movement can knock the larva off of the pipet tip. After many tries, I was finally able to get videos of whole-, half-, and quarter-sized larvae. These flow field images are informative of how well a given larva is able to produce localized currents, which are important in capturing food particles. Flow field images of whole, half- and quarter-sized larvae at 14 days old On Tuesday, the rough drafts of our posters were due. In our professional development session that day, we all projected our posters on the board and had a 10-minute rapid-fire critique session where everyone said what they did and didn’t like about each poster. This was definitely a bit intimidating, but it was very beneficial and it helped me see things that I wouldn’t have noticed myself.
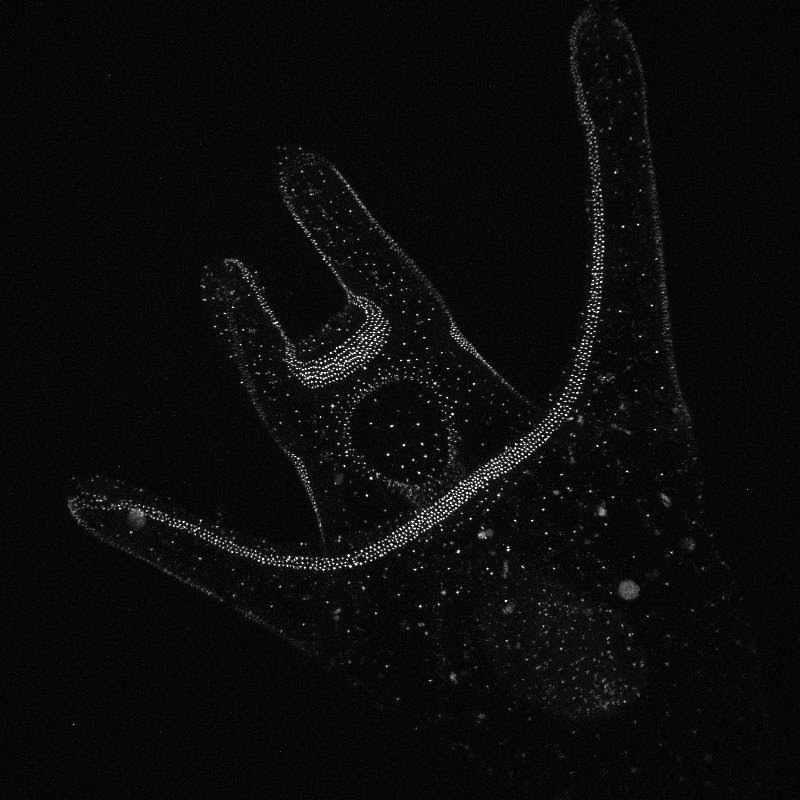
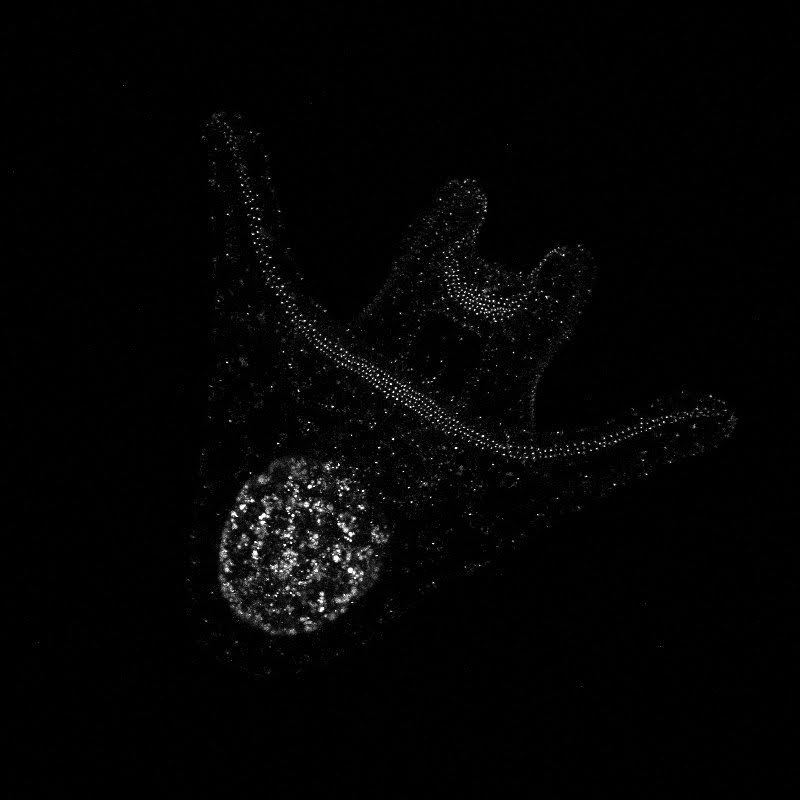
On Thursday, we got to attend the wonderful annual Invertebrate Ball! It was a busy week so, in all honesty, I most definitely slacked on my costume. However, Korrina was nice enough to lend me some “eel grass” (green ribbon) and a couple crab stickers before the event, so I was still able to walk the catwalk and participate in the festivities. The Invertebrate Ball was a great way to end a difficult week. Hello! It’s been a busy week here at OIMB – lots of late nights in the lab and cramming to collect all of our data before our posters are due. We’ve been documenting the development of our cultures of whole, half-, and quarter-sized sand dollar larvae, taking photos with the compound microscope every day. We also did antibody staining with some of the larvae that we had fixed in week 6, which should allow us to observe the nervous system under the confocal microscope and see if there are any observable differences between the wholes, halves, and quarters. The antibody staining was trickier than we expected. Since we didn’t have that many larvae to begin with (we can only separate so many blastomeres, and the war against bacteria continues) we didn’t want to sacrifice too many to the fate of fixation and staining. As a result, we only had a few larvae (anywhere from two to 14) floating around in the small vials in which they are preserved. To our surprise, when we went to do the antibody staining, we couldn’t even find half of our larvae! I don’t know where they went (our hypothesis is they traveled through a portal in the vial to another dimension), but spent a lot of time searching for these larvae that were M.I.A. In the end, we were able to save some, so here’s hoping that the staining process was successful and we’ll get some awesome pictures from the confocal! Centrin-labelled whole and quarter sand dollar larvae (left and right respectively) Note the ciliary band running along arms and around the mouth! In addition to antibody staining, we’ve been using high speed video to observe how effectively larvae are able to perform localized ciliary reversals and capture and retain food. This has involved a lot of sitting at the microscope, staring at a larva as it chows down on some yummy algae and trying to figure out which specific events are best to record. We just started two new cultures this past Wednesday and Thursday – we’ll be using the compound microscope to take measurements of arm length at different developmental stages throughout the next week, and we will also try to record more videos of feeding. We’ll be doing all of this on top of preparing our posters so that we can send them off to printing next Monday! A million things to do and not enough time, as usual.
On Thursday, we all presented our research to the public at the Charleston Marine Life Center. It was really fun to get to present our research to a broader audience and to brainstorm ways to explain things like blastomere separations to kids, teenagers, and adults. On Friday, we ventured to Eugene, where Dr. Kelly Sutherland gave us a tour of the science buildings at the University of Oregon main campus. We also visited the Museum of Natural and Cultural History and explored some of the shops downtown. It was a beautiful and sunny day in Eugene and a great way to end a moderately stressful week of research. |
AuthorHello! My name is Ana and I am a rising senior studying biology and music at the College of the Holy Cross in Worcester, Massachusetts. This summer, I am working under the mentorship of George von Dassow. I am looking forward to seeing where my research takes me and to becoming a part of the OIMB community! Archives
August 2018
Categories |
Proudly powered by Weebly










 RSS Feed
RSS Feed