|
Week 9 and I wish I had more time. The sea tables are dry, the chitons are free, and finally I don’t have at least one pair of socks soaked in seawater. This week I finished releasing my chitons with the help of Alondra and Nicole. We went out to Lighthouse Beach, North Cove, and South Cove. Putting the chitons back made me realize how well I had learned their specific habitat. I could look at a rock and think to myself, there are probably some chitons (C. dentienshere), lift a couple blades of algae and find several clinging to the rock. I guess you develop a knack for finding chitons when you’ve collected somewhere north of 200 of them! It was nice to enjoy the intertidal one last time before leaving for school in land locked Philadelphia. It has been neat to see the intertidal here and compare it to the intertidal near my hometown in Southern California. Seeing the differences in species and how species rich Oregon is has been so much fun. I think my favorite animal to have learned about (outside of chitons and echinoderms) in the intertidal here has been nermerteans (non-segmented worms, figure 1). I had never considered that worms would exist in the rocky intertidal but now I see them everywhere thanks to my fellow REUs Rebecca and Jacob (check out their blogs to learn more about these wicked-cool worms). I’m excited to see if I can find them in the places I go in Southern California this winter break. And so, as we face the final curtain, posters printed, and the final poster presentation (at the time of writing) inching closer I bid adieu and give thanks to Richard, Nicole, Maya, my lab mates (Alondra and Nick), and all the other REUs who made this summer memorable. And for you, the reader, I’m leaving you with pictures of many of the cool invertebrates I found this summer, enjoy and thanks for tuning in!
0 Comments
Its week 8 and the summer has been great! This week has been all about posters and trying to fit every piece of information I want to convey in a concise and clear manner in the space I have. To do so I’ve spent a lot of time in R and Photoshop, thinking (maybe too much) about statistical tests and models. It’s definitely helped my proficiency with coding and visual representation of my data! I have also; sadly, have begun taking down my sea tables and returning my adult chitons back to where I collected them. While the chiton exodus is sad for me, I get to enjoy the intertidal here a couple of last times. Which means (back by popular demand), more pictures of sculpins out of water (Figure 1)! Outside of making final poster edits and raising chitons, I have been able to enjoy my now limited time in Coos Bay. This weekend we went out to Hall Lake to enjoy the warmer weather inland. While we didn’t see a whole lot of aquatic invertebrates, the scenery was quite beautiful and dynamic. Right next to the lake was a sand dune with an incline close (or at least felt like it was) to 60 degrees, once you reached the top you could see ocean, sand dunes, and forest all in one shot (Figure 2).
Week seven and I named a chiton Devin (Figure 1). This week was the last series of low tides before I need to get my poster together. As a result, I have been spending my mornings in the tide pools collecting adults to spawn and my evenings taking pictures of larvae. I’m also doing experiments, organizing my data, focusing the narrative for my poster, and planning what final experiments I want to try. My last stint of experiments is going to focus on chitons’ ability to regrow their mysterious crystals. When I initially tired the boneless chicken water on my chiton larvae I found if left them in too long, the crystals would disappear or become noticeably reduced (in addition to calcium carbonate spicules). Originally an unfortunate byproduct, I want to take advantage of this phenomenon and demonstrate that chitons can grow these structures back. I observed this in two chitons when I first tried it but I want to demonstrate it more rigorously. This will provide direct evidence that chitons continually add to these structures. It will also allow me to see what conditions affect their ability to regrow them. Hopefully I can test the effect calcium has on their regrowth to compliment my other experiments, revealing if low calcium simply delays overall development or actually impacts crystal formation.
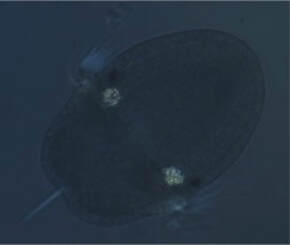
Outside of work I got to explore some of the greater Coos Bay area. Nick, Alondra, and I went out to the south fork of the Coos River. It was nice to get some sun, its most often cloudy here on the coast. The Southern Californian in me has needed some solar radiation! The river was quite beautiful and slippery. I certainly fell more times than I usually do in the intertidal, but it was worth it. We saw loads of crawdads and some newts, a change of pace from the usual chitons and urchins! Week six and I’m still learning new tricks —like how to make seawater fizz. This past week made me feel more like a chemist than a marine biologist, dissolving chiton shells and trying to take calcium out of real seawater. I started off the week by dissolving juvenile chiton shells in my boneless chicken water to see if the mysterious crystals persist the formation of the chitons’ shells. They do! We now know that chiton still have these crystals 15 days after settlement (22 days after hatching) and have a method to continue to observe the disco balls in the juveniles I have (Figure 1). I also redid my calcium experiment on a smaller scale, and this time my controls survived! However, they also did not form the spicules, which suggest the artificial seawater I am using has some unintended effect on chiton development. Additionally, my lowest concentration of calcium delayed the formation of the mysterious crystals by a day (relative to the other treatments), contrary to my previous iteration of the experiment where there was no delay. Therefore, I will be doing this experiment again to test the repeatability of this new result. Additionally, I will be modifying my methods to create calcium-depleted seawater from real seawater that will (hopefully) not interfere with the calcification of the spicules.
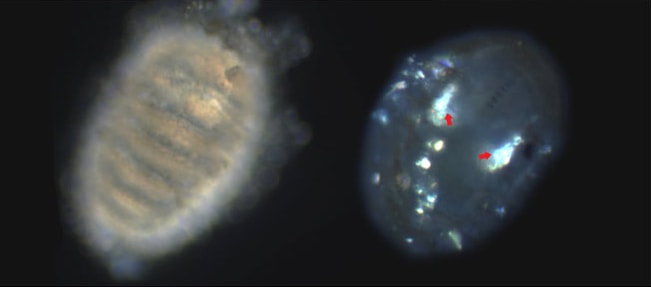
Unfortunately, I have been unable to find methods for removing calcium from seawater but many methods for making artificial seawater without calcium. However, I have been able to find some usable techniques, the most promising is making calcium oxalate from oxalic acid. In seawater, calcium and oxalate react to form a solid (calcium oxalate), allowing me to remove it from seawater. The unfortunate side effect is by putting oxalic acid in seawater, I acidify the water far beyond what could be considered normal for seawater. I have been experimenting with different ways to neutralize the acid before or after adding it to seawater. Playing chemist has been fun and led to some funny moments, a particularly memorable one is surmised by the quote, “Alondra, why is my seawater fizzing?” When I haven’t been dissolving shells and making seawater fizz, I’ve been out looking for chitons or relaxing with the other REUs at a bonfire. This week I started noticing a particular species of sea cucumber (Cucumaria miniata, Figure 2) at one of the sites I frequent in search of chitons. Richard had pointed them out to me last week at a different site so it has been fun to notice them on my own in a place I’m familiar with! Week five and my chitons are still alive –well, some of them at least. This past week I spent time taking advantage of good low tides to collect more adult chitons to spawn. I started doing some experiments with larvae I raised from their spawning. For my first experiment, I looked at whether calcium concentration affects the formation of the mysterious crystals. Across the different calcium concentrations (0.001, 0.0001, and 0.00001 Ca [normal seawater 0.01 M Ca) all larvae have their mysterious crystals and there didn’t seem to be an effect of Ca. Unfortunately, pesky ciliates infiltrated my controls so I lost nearly all my control larvae. I wish I had my controls, not only to make a relevant comparison, but also because my larvae in the other treatments did not grow their calcium carbonate spicules (Figure 1). This is interesting because spicule development usually precedes the formation of these crystals. It may mean calcium carbonate is not a part of the makeup of these crystals. However, I still am going to try a small-scale experiment to test the repeatability of what I saw (this time with controls that will live!). Additionally, I took a stab at putting larval chitons in acidified seawater (affectionately nicknamed “boneless chicken water”) to see if I can dissolve the spicules (calcium carbonate) without dissolving the mysterious crystals and keeping the animals alive. For the most part it seems to work, after four hours in the boneless chicken water, the chiton’s spicules are gone but the crystals appear to remain (Figure 2). The animals survived the treatment but after ~10 hours back in regular seawater, the crystals appear in scattered clumps within the animal and it is hard to be confident that what I see are actually the crystals. Regardless, this result is showing me that the crystals do not behave the same as the calcium carbonate spicules. This is a step in the right direction for what I am going to use boneless chicken water for: to dissolve the shells of juvenile chiton, which prevent crystal observation crystals. Hopefully, I can use this to see how long crystals persist after settlement. Outside of dissolving calcium carbonate and mourning the loss of replicated controls, I have started a collection of pictures with sculpins out of water. It is in its infancy but please enjoy some zany fishes!
Another week down and in the books! Our proposals and presentations were due this week so I spent a lot of time early this week fleshing those out. Writing my proposal definitely helped me clarify what my main goals are for this summer and the timeline I need in order to achieve them. Yikes, I realize how soon I’m going to have to leave OIMB; only four more weeks left!). I am focusing on how concentrations of Mg++ and Ca++ effect the formation of the mysterious crystals in my chiton larvae. In addition, I will be trying to observe how long these crystals last in chiton after they begin their transition into adulthood. As young chiton settle and metamorphose into adults, they grow their characteristic shells. This is a problem because the shell blocks the light needed to pass through the animal in order to see the mysterious crystals. So I will be trying a method (affectionately nicknamed “boneless chicken water”) to dissolve the animal’s shell while keeping them alive so I can observe the crystal. Its never been tried on chiton before so I will be adopting it for use with chiton! Outside of work I’ve been able to enjoy myself as well. Last weekend we had a bonfire and got to see some fireworks for the Fourth of July. There has also been a series of low tides so I’ve gone out and searched for chiton to spawn for my experiments and along the way I’ve seen some pretty neat animals and phenomena. A personal highlight was seeing sculpin (an intertidal fish) hanging out in algae –a foot above the water line. These fish are amphibious and can survive in the moist habitat left behind by the retreating waters during a low tide! I’ve also seen some nudibranchs (sea slugs) and giant sea stars.
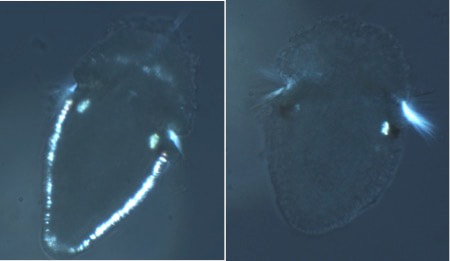
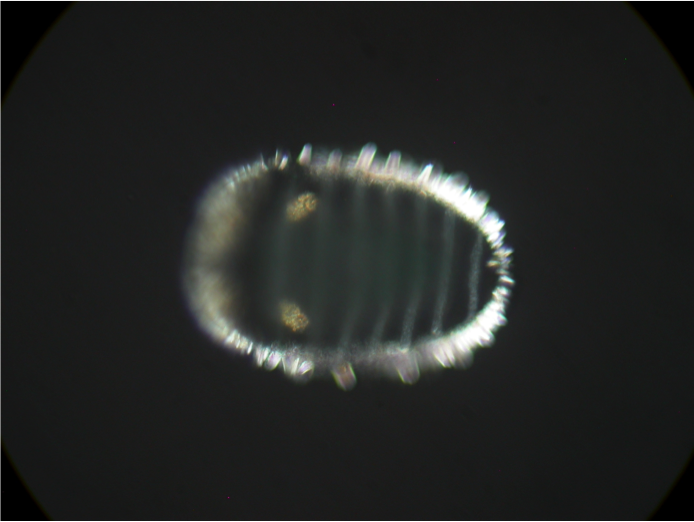
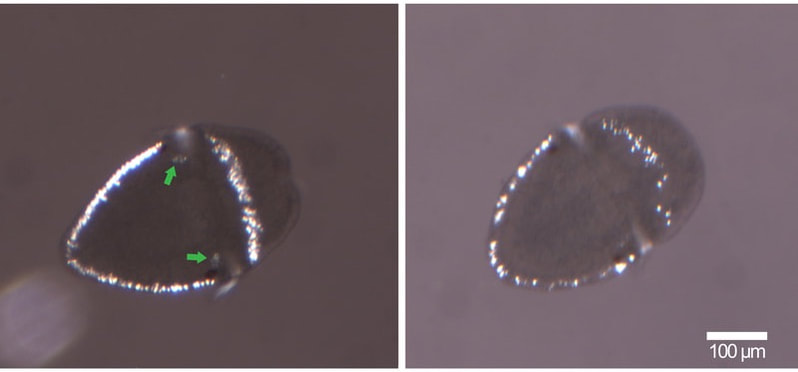 figure 1. Two chitons, grown at different salinities: normal on the left, and low salinity on the right. If you look closely at the spicules (the white spots that form a band around the animal), you can see the ones on the left are more developed than the ones on the right despite these animals being the same age. You can also see the crystals in the chiton on the left, they appear as two faint white dots and are indicated by green arrows. figure 1. Two chitons, grown at different salinities: normal on the left, and low salinity on the right. If you look closely at the spicules (the white spots that form a band around the animal), you can see the ones on the left are more developed than the ones on the right despite these animals being the same age. You can also see the crystals in the chiton on the left, they appear as two faint white dots and are indicated by green arrows. Wow, week three has come and gone faster than you can say Cyanoplax dentiens! This week I set up an experiment testing how larval chitons respond to different salinities of seawater. Manipulating the salinity of seawater changes the concentration of different ions (such as calcium or magnesium) available for animals to build shells or other crystalline structures. So I hypothesized decreasing salinity (and therefore the concentration of ions needed to build different crystals) would delay the formation of the mysterious crystals. To do this I had to place the chiton in different salinities before these crystals formed. And, as it turns out, I did manage to delay the formation of crystals –but that’s not the full story. Unfortunately (but also interestingly), chitons at different salinity levels (low and normal) ended up on different developmental timelines. As chiton larvae grow, they add new structures, which allow us to mark where they are in their development. Right before chitons develop the mysterious crystals, they grow gridle spicules (calcium structures that form around the periphery of the animal). In my experiment, chiton in normal salinity formed spicules before chiton growing in lower salinity seawater, despite being the same age (figure 1). The formation of spicules occurs right before the mysterious crystals so while I was able to delay their formation it was likely an indirect consequence of delayed development rather than a direct effect on the crystals. Therefore, my future experiments will manipulate ions (e.g. calcium) without changing salinity and will focus on trying to target chitons in the brief time when they have spicules and no crystals.  figure 2. A gothic looking clump of gooseneck barnacles, usually these animals are off-white and with black cirri (the appendages barnacles use to feed). These animals are gray with red cirri, crazy! figure 2. A gothic looking clump of gooseneck barnacles, usually these animals are off-white and with black cirri (the appendages barnacles use to feed). These animals are gray with red cirri, crazy! Outside of running experiments, this week we went camping at Sunset Bay and trekked to an island at low tide. The tide was low enough where we could wade to the island and poke around in the tide pools. We saw plenty of awesome stuff but my highlights were: finding a red urchin test (shell) about the size of a grapefruit, seeing a neat color morph of a gooseneck barnacle (figure 2), and coming across dislodged sea palm (figure 3). Sea palms are pretty intense algae because they usually exist only in areas exposed to the full force of crashing waves.  figure 3. The picture on the right is the dislodged sea palm I found. The left? Home sweet, sea palm home! Within the green circle is a cluster of sea palms, these algae are known for inhabiting areas in the intertidal where the rocks experience the full force of crashing waves –and they seem to like it! 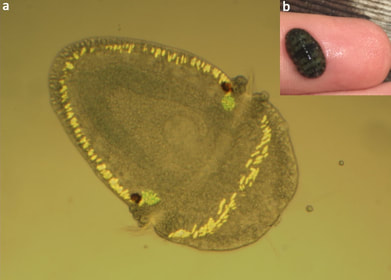 a. A larval chiton, Cyanoplax dentiens, under a microscope (~ 0.4 mm long). The mysterious crystals are highlighted by a fluorescent green stain and are located next to the larval eyes (red spots). The bright yellow structures are spicules forming in the girdle b. An adult Cyanoplax chiton on my finger for scale! a. A larval chiton, Cyanoplax dentiens, under a microscope (~ 0.4 mm long). The mysterious crystals are highlighted by a fluorescent green stain and are located next to the larval eyes (red spots). The bright yellow structures are spicules forming in the girdle b. An adult Cyanoplax chiton on my finger for scale! Hi my name is Andrew, I got my luggage (finally), my vaccine, and I’m ready to go! Originally from sunny San Diego, I’ve been studying Biology in landlocked Pennsylvania at Villanova University for the past three years, so I am excited to be back on the West Coast (Best Coast) and doing research at OIMB this summer! At Villanova I work in a lab focusing on the Pacific purple urchin, Strongylocentrotus purpuratus, and their adhesive tube feet. I am a big sea urchin nerd, but this summer I will be trading calcite spines for aragonite shells and working with chitons. I will be working in Dr. Emlet’s lab studying their larval development. In particular, I will be focusing on mysterious crystals that form near and possibly within the larval kidneys. Many animals have two sets of kidneys during their lifetime, and chiton are no exception. The larval kidney only exists for a short time, which may mean these crystals are associated with their failure or simply chiton kidney stones! But all this is a bunch of maybes, we really don’t know much about these crystals. My work will focus on how chemical environments (e.g. different salinity) affect the growth and appearance (or disappearance) of these crystals. I will also look into what these crystals are made of. I am excited to work with a new animal and life history stage and am already feeling the call to chiton nerdy-ness as well. I have really enjoyed the first two weeks at OIMB and meeting all the other REU students. In the Emlet lab there are two other REU students, Alondra and Nick, working on juvenile sand dollars and barnacle larvae, respectively. It’s been great to go towing for plankton and discussing experimental design with them. I am looking forward to going on more trips to look for chitons with them and learning more about their projects. A really special moment for me this week was seeing purple urchins at the field sites of Dr. Tom Ebert, who mentored my professor at Villanova. I had only ever read about the site and to see it in-person with its relevance to my research experience was pretty profound. |
AuthorI'm a rising fourth year at Villanova University in Pennsylvania from San Diego, California. I enjoy going tidepooling and making music. I'm looking forward to a great summer at OIMB! Archives
August 2021
Categories |
Proudly powered by Weebly



















 RSS Feed
RSS Feed