|
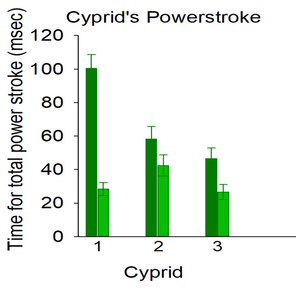
The final week of the REU internship has come and gone, these nine weeks have flown by. Doing research everyday with a group of wonderful people is something I will treasure and fondly look back on as I progress toward my career goals. With this being the last week of the internship most of my research had already reached a stopping point meaning I had all week to work on my poster. While I had all the data and words needed for my poster by Monday, the actual arrangement of pictures, data and words took the rest of the week for me to figure out the placement which would be most pleasing to follow for the eyes. While working on my poster, I found three key outcomes. I found one after I ran 3 paired t-test for my data on the time per power stroke. These results show the powerstroke happening over a shorter time frame after cutting the midline setules. I also found significant evidence of faster speeds of particles in the vicinity of the moving setae after cutting midline setules compared to before cutting the setules. Which suggest faster setae speeds after cutting the midline setules While these show some change in behavior, the physical chance that results from cutting the midline setules, is only partial opening of the setal fan during the powerstroke. When the setules are not severed the setal fan opens fully with the exopod and endopod spread wide. However when I cut the midline setule the endopods on the paired legs stayed closed while the exopods continued to spread. This outcome was not what I expected because I thought it would be a full open fan or not open at all, however seeing the partial open fan has given me new ideas for father research on the setule functions. One thing I’d like to research farther on setules functionality is severing the setules between a legs endopod and exopod to see if this causes on exopod to open by itself while the two endopods and the other exopod try opening in a full fan during the powerstroke.
0 Comments
Welcome back, my week started off fantastic. It happened to be my birthday over the weekend so many of the REU interns had dinner at 7 Devil’s to celebrate. We also went to Hall Lake since you can swim in it and some of us have been dying to get in the water. After the weekend it was time to analyze all the videos I recorded of cyprids swimming this summer. After reviewing many videos I narrowed them down to only those which had the same cyprid before and after cutting the midline setules. I watched the videos frame by frame to track when the powerstroke of the cyprid starts and ends. Then I recorded the time a powerstroke takes, as well as the speed of any particle that stays in the same plane of view while the powerstroke happens. Once I organized my data on spreadsheets, I was able to plot graphs to compare the difference between the time per powerstroke and particle speed from before and after cutting the cyprids midline. With just the raw data I could not see much change in time per powerstroke or particle speed, however once I made the graphs there was a significant difference in time per powerstroke with the non-cut powerstroke taking up to 3 times as long as the cut powerstroke. There could have also been a difference in particle speed as well but since the standard error bars cross over I will have to examine more particles before and after cutting the setules on cyprids. After I finished collecting and sorting my data, I started working on my research poster. My poster took much longer than anticipated. All week I was cutting as many words as possible without losing the purpose behind the research I did. I also worked a lot in photoshop this week since much of my work required visual aid for a better understanding of the changes I made to the cyprid during its powerstroke. I am looking forward to presenting my poster with my results this upcoming week
Welcome back, this weekend I got to enjoy the sun again. Andrew, Alondra, and I decided to go upriver to catch some sun and see some freshwater animals. While there we saw plenty of small fish, salamanders, and even a yellow crawfish (all others we saw were a shade of orange). After leaving the river we were greeted with a blanket of fog on the coast which has stayed the rest of the week, so I’ve been grateful for the sunny weekends. This week I started off sorting through all the videos I made in past weeks of cutting my cyprids on a stick. If you want more information on how I make my cyprids on a stick or how I cut them, I talk about it in more detail in past blog posts. I sorted the videos first by what day they are from, then whether they are from before or after I cut setules. After I got my videos in order I began to count the time it took for each powerstroke. I was able to do this using a program called ImageJ which turned each video into a collection of stacked images allowing me to see the cyprids movements during each frame. Since the videos were taken in 2000 frames per second I know each frame takes place over 0.5 millisecond intervals allowing me to calculate the amount of time each beat took by counting the frames it took for each powerstroke. I hypothesize that the power stroke should be faster if the setules are cut because the setal fan will experience less drag. I was also tracking particles in the water to examine if the particle velocity changes with cutting the setule. I hypothesize that cutting the median setules will result in setae being less spread apart and so less water will be moved on the powerstroke. After examination of the calculated particle velocities I saw they varied between particles even during the same powerstroke. This is related to their proximity to the setae and speed that setae are moving. However the time per powerstroke seems to indicate that once the setule is severed the powerstroke will finish its stroke sooner than one with its setules connected. But I still need to make more cuts to increase my sample size to make sure it’s not just the individuals I cut.
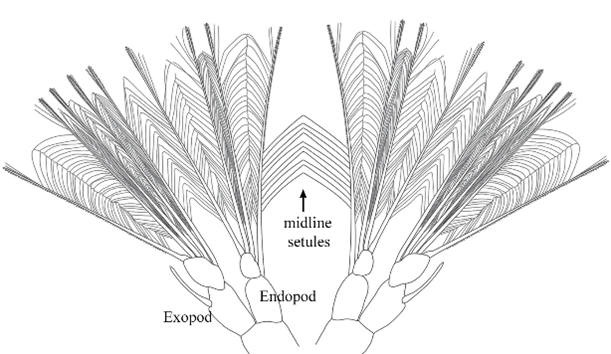
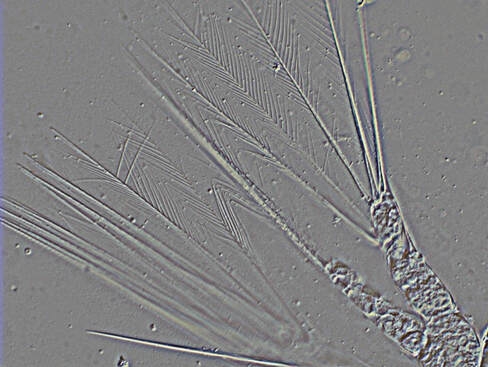
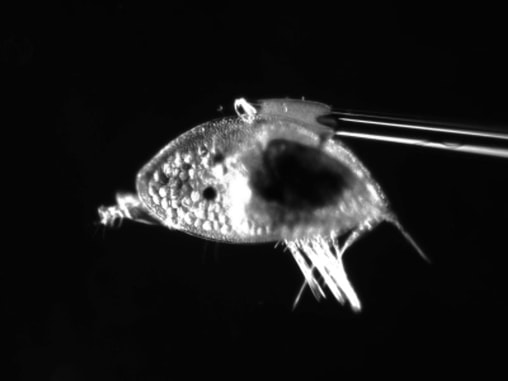
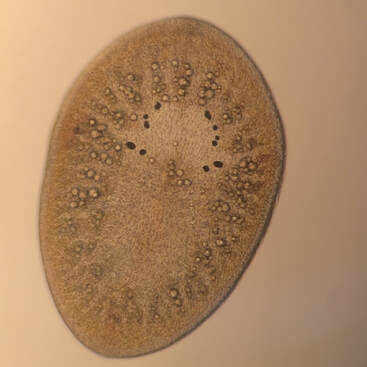
Hello there, over the weekend Jacob and I went on a trip up the Coos River in search of sun. This ended in success with us finding a nice spot in the river to relax all day and give Jacob the sun he desperately needed. While basking in the sun we had an eagle glide over us unphased by our presence. We also saw some juvenile salmon while there which is always nice to see though they run when you try to get close enough to see the type. My research on how cyprids, the final larval stage of barnacles, swim progresses. Cyprids use hairy bristled appendages to swim. The weird part about a cyprid’s bristles (setae) is that the setae are linked to one another by setule, the hairy part of the appendage. (see Fig 1) On every other day I conducted plankton tows during high tide to capture my cyprids. I collected more often because diatoms were abundant resulting in few cyprids per tow. While I was isolating my cyprids from the plankton I didn’t find anything too unique. I did find out the nemertean from last week’s tow was in its larval stage. After isolating my cyprids, I made some “cyprids on a stick” (see Fig 2) by super gluing cyprids to a micropipette. I then took High speed videos of the cyprid with and without particles in the water. I positioned the cyprid to have its setae swimming toward the camera to capture a better view of the setules connecting the pairs of legs before and after cutting them. I go into more details on how I take the video and the process of making my cyprids on a stick in previous post. This week I tried a small knife tip instead of a pulled micropipette to cut the setules. I think I had some success with this and caused less damage to the other setules but it is hard to tell with just one cyprid. The result of cutting the setules only in between the two appendages seemed to be partial opening of the outer part of the fan and no opening on the setae close to the severed setules. It also seemed that the beginning of the powerstroke was faster but the end was slower resulting in an overall slower powerstroke for animals with severed setules (compared to ones with intact setules) but I can’t say for certain until I look at the videos frame by frame in ImageJ. I have seen some consistency with the results in the 3 cyprids I have cut however I still need more data in case I am causing more damage than just cutting the setules. The results seem to suggest the more setules that break the less open the fan becomes on the powerstroke. Also without the linkage between the paired appendages, the appendages lose their sequential order when grouping together at the end of the powerstoke.
Hello again, this week started with a fun tidepool excursion during low tide on the weekend. I went to Sunset Bay with Jacob and Andrew in search of critters hidden amongst the rocks and algae. Andrew went sea urchin hunting while Jacob and I looked in different puddles filled with a variety of animals. The coolest of which being a long bright red nemertean (Tubulanus rubra). I also noticed gray coating on many of the muscles and bare rocks which are thousands of barnacle juveniles! My research on how cyprids (the final larval stage of barnacles) swim continues. Cyprids use fuzzy bristled appendages to swim. The unique thing about cyprids bristles (setae) is that they are linked to one another by setules, the fuzzy part of the appendages (see Fig 1). On Monday and Thursday, I did some plankton tows during high tide to capture my cyprids. In the plankton sample I found a small nemertean, roundworm without segmentation, swimming through the other plankton. While looking at the other organisms I isolated all the cyprids I found. This past week after isolating the cyprids, I super glued the cyprids on a pulled micropipette - making “cyprids on a stick.” I then made highspeed videos of my cyprids swimming while held in place by the needle they are glued to (see Fig 2). After an initial video of the intact cyprid, I tried to cut the medial setules connecting the appendages across the midline. I did this by waiting for cyprids to swim and then quickly moving a micropipette through the region where these links are. I predicted that if these setules were cut, the setae on each leg would no longer spread apart on the power stroke. Whenever I got a clean cut, I recorded another video to see if the setae on the legs were still spread apart or no still bundled. After a few attempts I was able to successfully sever some of the legs. That makes it two weeks in a row with a complete or partial cut of the setules, and that is pretty exciting! The setae of appendages which appeared to have severed setules did not open in a fan during the powerstroke, instead the setae remain bundled while the legs moved apart though their power stroke. These results suggest the medial setules connecting the two pairs of appendages are used keep the medial setae joined and this helps spread the setae apart while muscle contraction moves the legs apart. Though more videos on different cyprids will have to be done before any actual conclusions can be made, I’m getting useful data. It’s difficult to verify that the setules have been severed and that the larvae were not just acting strangely. The only way to do this is to dissect the thorax with the swimming appendages and try to spread out the setae to examine the setular linkage. This is problematic in that the links may be broken in the process of spreading the setae. Many cyprids lost their lives in the process of learning how to place the coverslip right, and I’m still working on getting better at it. I’m excited to see if the results for severing the setule will be the same with different individuals and getting better at viewing the setules up close.
Hello again, we started out the week with a huge bang as Fourth of July was last Sunday. I am doing research on how cyprids, the final larval stage of barnacles, swim. Like other small crustaceans cyprids use bristled appendages to swim. The unique thing about cyprid swimming bristles (setae) is that their setules (tiny hairs on the bristles) are linked to setules on adjacent bristles. I am examining how water and particles flow through and around these appendages. I am also doing research on how severing the connection between the pairs of appendages affects the opening of the fan. I started the week with another plankton tow. In the plankton sample I found a small jellyfish that is the reproductive stage of benthic hydroids (tiny coral like animals. While looking at all the organisms in the plankton I was able to isolate all the cyprids too. After I isolated my cyprids, I went to work putting them on a micropipette. I achieved this by dropping one on a Chem wipe then positioning it so that I can tap it with the tip of a pulled micropipette that has a drop of super glue on it. I position the cyprid on the micropipette so I can capture a video of the cyprids swimming pattern with little interfere from the pipette or the glue. After I have enough cyprids on a stick I can start recording my cyprids in 2000 frames per second (normal video is in 30 frames per second). First, I record a lateral view of the cyprid swimming in place. I then added algal cells to the water to be markers for water movement through the pairs of setae and the setules between the setae. Then I record a ventral view of the swimming cyprid with the legs coming toward the camera to show a different angle of the swimming motion. After getting the videos of the cyprid on a stick, I started the process of cutting the setules connecting the pairs of setae. This was done with a pulled micropipette on the end of a micromanipulator. I would wait for the cyprid to start swimming then place the pulled micropipette in between the paired legs and swiftly pull up to cut the setules along the midline that link the left and right legs. After accidentally jabbing a few cyprids, I figured out a technique that allowed me to cut most of the setules connecting the paired leg. Success has never felt so sweet since cyprid were alive and able to swim after the cut! Since the cyprid was still alive and trying to swim I quickly recorded a video of the cyprid swimming with the setae facing toward the camera. As I watched the video, I noticed that the fan wasn’t opening up nearly as far as with the setules connected, but it was still able to open them a little. After keeping the cyprid in seawater for a day I took another video that showed the cyprid going through the swimming motions, but without opening its legs into the fan-like structure for the powerstroke. These changes through time suggest that the cyprid was aware that its legs had been damaged and did not try to open them. So now I just have to cut the setules on cyprids many more times to try to figure how they help the animals swim.
Welcome back! We started off the week with a boat trip in the bay since the swells were to high in the ocean for an enjoyable ride out there. We did a few otter trawls at different locations in the bay to get familiar with the creatures living in the benthic layer of the bay. Resulting in me seeing Richard throw crabs, unintentionally, at another REU intern since he was a man on a mission emptying the otter trawl on to the boat. Then while waiting for the otter trawl we saw seagulls impeding on a Bald Eagle’s freedom, by chasing the eagle while the eagle was just gliding majestically. I am researching swimming in cyprids, the last larval stage of barnacles. To swim cyprids use 6 pairs of bristle-covered legs and on each leg pair the bristles are interconnected forming a mesh like fan. I am researching how water and particles move through and around these structures before and after severing the connection between the leg-pairs. The real work begins with plankton tows every other day. Plankton nets are basically cylindrical underwater sock-like filters (that have a bottle at the toe end) pulled through the water by a rope attached to the open end. The abundance of cyprids found in my plankton tows varies due to many factors (winds, tidal stage, water mass) that may lead to great variation between tows. After I isolated some cyprids from the rest of the plankton, I started practicing attaching a cyprid to a micropipette. This might sound easy enough but lightly brushing the cyprid while it’s out of water makes it hop like a flea, also trying to use as little superglue as possible. It took me the whole week to get better than a 25% success rate in putting my “cyprids on a stick” without gluing their legs closed. Another hurdle is tethering the cyprid in the right orientation so you can get a video with the legs in focus.
Once I became comfortable with tethering, I ran into the problem of trying to get videos of something that cannot communicate with you. While I can have the camera on the cyprid ready to record, the actual length it takes for one powerstroke and recovery stroke is shorter than it takes me to react, so I hope it beats more than once so I can capture a video of its motion in 2000 frames per second! (normal video is 30 frames/s) So now I’ve got my cyprids on a stick set up for video, and I’m trying to sever the connections between the leg pairs. The plan is to move a micropipette (held by a micromanipulator) into position near the legs of a resting cyprid, wait for the cyprid to swim and then move the pipette in a manipulator-controlled, sweeping action to break the leg connections. So far, I have only been able to jab the legs or spear the cyprid because I must position a micropipette very precisely. I am hoping I can time everything perfectly. All of these challenges have helped me gain a steady hand and to carefully think before gluing the cyprid, so I look forward to the things I learn from the next hurdles thrown my way. Hello Readers! My name is Nick Nodzak and I was born and raised around Atlanta Georgia. I enjoy being around nature and pushing myself to new experiences. These two loves helped to draw me from Atlanta and let me experience the diversity of people and cultures around the United States, with many leaving lasting impressions and shaping the person I have become. I am currently attending the University of Oregon in the pursuit of a bachelor’s in marine biology, so living on the coast right next to the dock and shore makes getting live specimen more of a treasure hunt, which I have enjoyed, with a scenic mountain backdrop. I am working in Dr. Richard Emlet’s lab with fellow REU interns Alondra and Andrew. We are all focusing on different projects with Alondra “tanning’’ sand dollars, and Andrew hunting for kidney stones in chitons. Even though we all are working on different projects, having them in my Lab has been wonderful, the amount of ideas bouncing between us to help one of our projects and the synergy between us has amazed me since it has happened so quickly. In my research I will be studying swimming in cyprids, the last larval stage of barnacles.
Cyprids are tiny (<1mm), nonfeeding, bean-shaped “bugs” whose job is to swim fast and then cement themselves down. They swim with 6 pairs of bristle-covered legs that are unusual because the bristles are linked along each leg and between leg pairs. This linkage results in a fan- shaped structure when bristles (setae) are spread to move water on the powerstroke. I will be looking at the functional consequences of this linkage. I plan to sever one or more of these linkages to see if this changes how the fan opens. I also hope to examine how particles and water move through and around these meshed paddles. My work will help explain how this newly discovered linkage works. I am excited about this research because I am learning about larvae, how to manipulate and experiment on animals under a microscope, how to use a highspeed video system, and explore fluid moving around tiny organisms. |
Proudly powered by Weebly