 I can’t believe that this was the last week of the internship! The summer went by very quickly and I still have so much that I want to do! This week I continued to take videos and pictures of the embryos. Since I had collected all the data I needed, I was able to try some different methods that could possibly make the process easier. We tried to soften the egg shell to see if it made the eggs easier to inject and easier to put on a slide. The eggs did remain easier to inject for a longer period of time, but they were very delicate and didn’t enjoy getting stuck with a needle. Everything on the inside of the cell tended to pour out when the egg got poked with a needle. However, the eggs were considerably easier to flatten on a slide. Usually the egg shell is so firm that it gets broken before it gets compressed. On Thursday I tried injecting Nina’s probe into some barnacle eggs. Since it lights up so well and clearly showed part of the cell in starfish embryos, we though that it was worth a try in barnacle embryos. While the probe was expressed (It light up) in the barnacle embryo it didn’t attach specifically to anything in the embryo. So, it was there but it didn’t show us anything useful. On Friday we had the poster session. I enjoyed getting to share what I had learned with all the people who walked through. A condensed version of my poster is that my research is on barnacle embryos and how cell polarization effects the cleavage furrow pattern. I injected different RNA or protein probes into the embryos and then used a confocal microscope to track the probes in the cell. I discovered that barnacle embryos have an asymmetrical first division that results in one large and one small cell. This division is related to cell polarization. Before first division the cell’s contents are polarized into granular yolk and globular yolk. After the cell divides there is a small cell that contains mostly granular yolk and a large cell that contains most of the globular yolk and a little of the granular yolk. The way that the barnacle embryo accomplishes this is by using two cleavage furrows. The primary furrow forms down the center of the embryo and then a second medial furrow forms and meets the primary furrow in the middle. The cell continues to have this polarization in the later stages. The small cell divides into approximately equal daughter cells but the large cell divides into a large cell that contains mostly globular yolk and a smaller cell that contains mostly granular yolk. The result is three approximately equal small cells and one large cell. I have really enjoyed this internship. It was incredible to be able to learn how to do research and then to work on a project. The skills that I learned are ones that I know will be able to apply later in my career. The professional development sessions have given me more knowledge to use as I continue with my education. Thank you, Dr. Watts, Dr. Emlet, Nicole, and Dr. von Dassow for this incredible opportunity. I also enjoyed getting to spend time with all the other interns and I really enjoyed getting to work with Philip and Nina in the lab. Thanks for the awesome summer everyone!
0 Comments
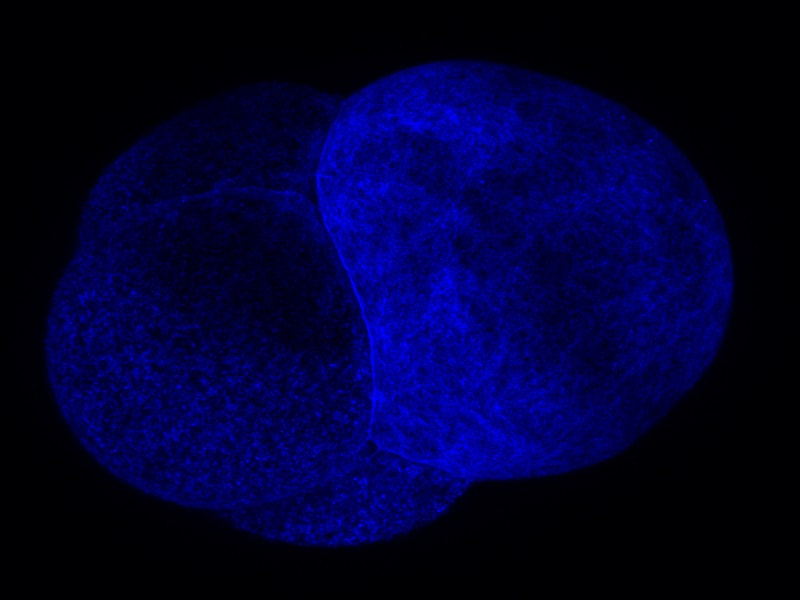
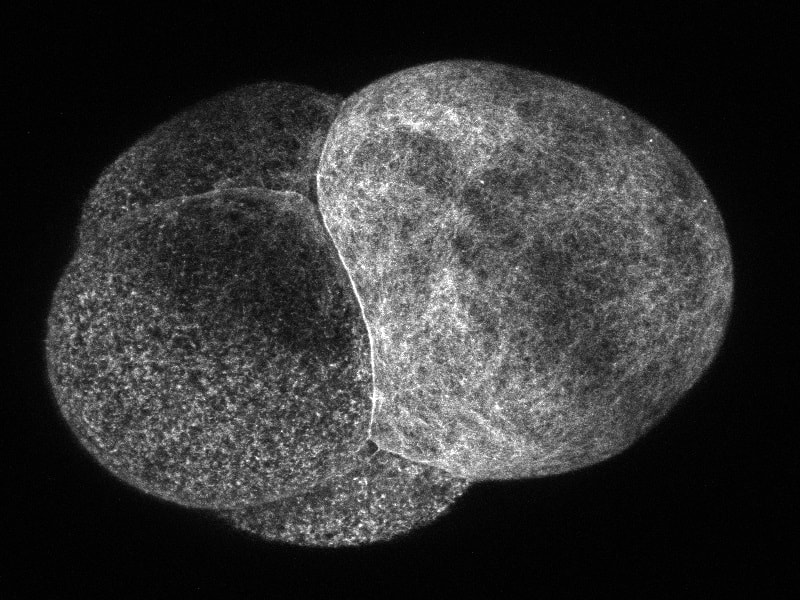
Welcome to the week 8 post everyone! This week we’ve been doing a lot of work on our posters. On Tuesday we had a poster critique among the interns and Dr. Watts. We projected everyone’s posters up on the screen one at a time and gave feedback on what we liked, or thought could be changed. I greatly appreciated the advice. It was fascinating to see everyone’s posters taking shape. Then we turned the posters in for the final review on Friday. It has been surprisingly difficult to cram everything I have seen and observed onto one poster. The most constant comment I got at the critique was to get rid of a lot of the text! Trying to condense everything has made me sift through what I have observed and try to pick out the most important things. Probably the most important finding that we observed was a secondary cleavage furrow. The first and main cleavage furrow starts out like it is going to divide the embryo into two equally sized cells. Then a second furrow forms on the side and continues moving towards the center until it connects to the main cleavage furrow. The result is that there is one large cell and one small cell. The other thing I’ve been doing is sorting through videos and pictures. Dr. von Dassow taught me how to use some basic functions with ImageJ Fiji. The program is incredible, and I’ve had fun turning different probes into different colors to emphasize different pieces of the embryo. Normally we work with grayscale images because that is the best color range to pick out details in. Greens also work well but blue doesn’t show details at all. And while some color combinations, like green and magenta, clash they actually show off the different parts to the maximum advantage. I’m going to attach some images so everyone can see the difference. While I have enjoyed getting to look through the data and explore ImageJ I’m always glad when I have time to inject and image more embryos. Since I have all the data, I need I have been able to some new ways of setting things up and see what else I can learn with out the pressure of getting a result. On Friday we tried putting the embryos in a chemical that keeps their egg shell soft and pliable. Unfortunately it is a lot harder to get the embryos to stick to the injection plate while using 3 APA but we’re still working on it. Below you can see the pictures of the embryos, they are all the same brightness but you can see the details on the cell surface much better in the gray than the blue. Welcome to the week 7 blog post everyone! This week Dr. von Dassow was gone for a conference and Monday was a bit of a struggle. I quickly found that I can’t take 15 minutes to get a needle loaded if I want to get a plate of embryos injected before they get too hard! However, by Tuesday I had figured out how to time everything. I also got a lot faster at doing everything which helped a lot! I spent this week collecting data and trying to learn everything I can by watching the embryos. This Thursday yielded some exciting results for me. I think I’ve found a way to successfully predict where the smaller cell will divide from. Before the embryo divides a layer of something containing the utrophin probe sweeps across the surface of the embryo. What I’ve found from taking and watching videos is that the last spot to be covered by this “hood” will most likely be directly opposite where the smaller cell will be. So, by using a utrophin probe and an EB1 probe that tracks the tips of microtubules, I was able to get a couple of videos showing the cleavage furrow in one day. So instead of getting 10 videos with only 1 or 2 showing the cleavage furrow, I got 2 out of 2 videos showing the cleavage furrow. I have attached the first video that I got on Thursday below so you can look at it. I was extremely excited when I was watching it! If I’m right about the importance of the hood than it will be much easier to find embryos in the correct orientation to film. However, right now the only way I can see the hood and tell which way it is moving is by taking about 3 minutes of video. This works well if the second or third embryo I look at is oriented the right way, but I only have a certain amount of time between when the hood starts forming and when the cell divides. So, I only have time to check a few embryos before they start dividing. I’ll work on testing this idea and hopefully I won’t be getting any more videos that are looking at the back of a dividing embryo! On Wednesday I got to watch two interesting presentations. The first was a defense of a marine biology honors thesis by Alexander Hethman. I’ve never watched a defense of a project before and the topic was interesting. Alexander knew a lot his topic and I really enjoyed getting to hear his defense. We also had Dr. James McClintock speak for our seminar. Dr. McClintock has been doing research in Antarctica and told us about the effects of climate change on Antarctica. He has personally observed changes in Antarctica and showed diagrams of the receding ice sheets and glaciers. He also had some funny stories to tell about his time in Antarctica and as he spoke to others about climate change. On Saturday we will be spending a few hours in the CMLC tell the people who visit the museum about our projects. It should be a lot of fun.
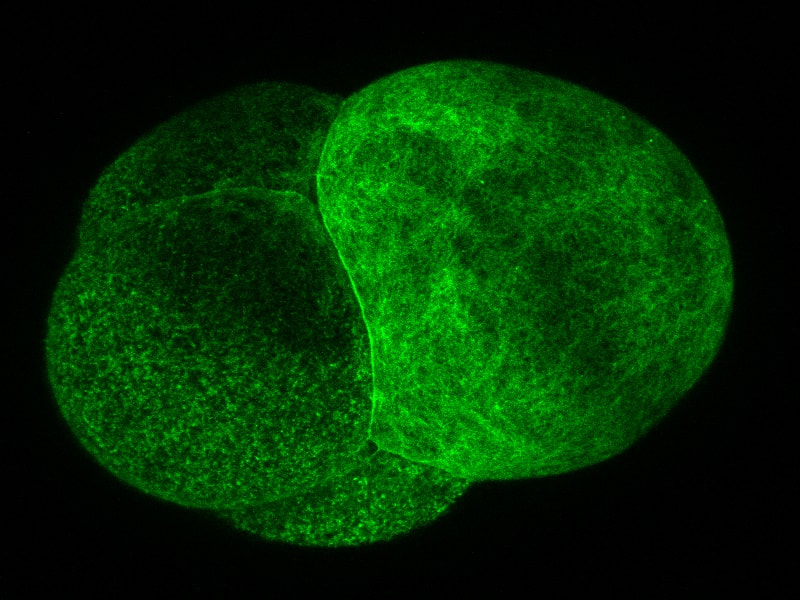
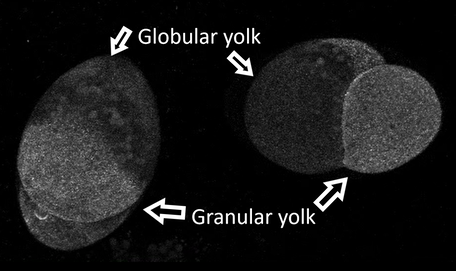 The Differentiated Yolks The Differentiated Yolks Welcome to the week 6 blog post! Because the aPKC probe I was originally planning to use didn’t work, I have been working my way through a few other probes. I need to use a probe that allows me to see the structure of the cell and the cleavage furrow. On Monday, I tried two different kinds of myosin probes. Interestingly one of these probes was formed by using antibodies from camels! Although one of the probes was visible, it wasn’t particularly bright or very helpful. So, on Tuesday we tried a utrophin probe. The probe has been used before in the lab so we knew it would work. The probe lets us see the cleavage furrow, and if the spindle poles are close to the surface, we can see them too. We have been trying to get videos of the embryo dividing during first cleavage, but the success rate is very low right now. Let me rephrase that. We have plenty of videos of the embryos dividing, you just can't see the cleavage furrow because the embryo rotated around to hide the division! If I can see the spindle poles I can guess where the cell will divide. However, in barnacle embryos the first division is not symmetrical. The embryo divides into two cells; the first is small and contains the granular yolk and the second is large and contains a little of the granular yolk but also contains all the globular yolk. In the picture I have pointed out the two types, but you can also see the different sizes of the cells. So even though we typically know where the spindle poles are, we don’t know if the top quarter of the cell will become the smaller cell or the bottom quarter. So far it has been frustratingly hard to find a cell the divides where we can see the cleavage furrow. So, part of what I am doing now is looking at videos that have been taken and taking pictures of embryos in different positions so hopefully we can find a pattern that will let us choose the best embryos to view. While the video can only capture what is happening on and right below the surface of the side of the embryo closest the camera, the pictures let us see about half way through the embryo. The picture below is one of the ones I took on Thursday using the utrophin probe. Hi everyone! This week I finished my aPKC GFP probe and tested it for the first time on Thursday! It has been incredible to work through all the steps necessary to create a probe and then to end up with something that could be tested. One part of being a scientist that I am discovering is that sometimes what you are testing works perfectly with your hypothesis; but other times you can end up with a result that doesn't. While the probe did express on Thursday it didn’t begin to show up visibly until the eight-cell stage and even then, the probe was hazy. We were hoping to see it clearly between cells. The two places that the probe did show up clearly were in the nucleus and in small circles that we think could be vesicles. Unfortunately, neither of those were places where we wanted to see it! So, the next step we tried was to increase the amount of RNA that we inject into the embryos. On Friday we injected three times as much RNA as we did the day before. The embryos seemed to handle the higher dosage just fine, but the same dimness and lack of defined location in the cell was still observed and the probe was still not well defined by the time that cell polarization or first cleavage happened. However, when we looked at the cells that we injected on Thursday the probe was working. We could see it in the cell junctions. So, the probe is working, it just isn’t working at the stage in cell development that I need it to. There is a possibility that the probe only shows up briefly during first cleavage, so we just aren’t catching it when it is lighting up. I will be continuing to take videos of the injected embryos to see if we can see anything happening.
This week I thought that I would show you what I am looking for when I am watching the barnacles. Image 1 is a barnacle searching for a mate. The long thin projection is the penis that he will probe the other barnacles with as he tries to find a barnacle close to him to fertilize. Image 2 is a successful fertilization. You can actually see the thin thread of sperm as it moves from the donating barnacle to the receiving barnacle. Image 3 shows another way to tell if a barnacle has been fertilized. It will release a cloud of excess sperm 10-20 minutes after fertilization. The pink is from the colored pencil that we use to keep track of the barnacles. After I see a mating or a release of sperm I wait until the animal’s feeding appendage is out again before retrieving the brood. Then I pull apart the brood so I can inject the embryos.  Hi everyone, we are now on week 4! This week I have seen some awesome things in the lab. On Thursday I injected a new probe into barnacle embryos. It wasn’t the probe I have been working on creating, but it was one that Dr. von Dassow wanted to test. This probe attaches to histones, which are what the DNA is wound around in the chromosomes. We wanted to see if we could get the probes to change from emitting green light to red light when we shone a violet laser on them. It took awhile of playing around with the laser to get the protein to change color but in the end, it worked! This could let us track if a certain cell always makes the same part of an barnacle. An update on my research project is that the probe is getting close to being finished. I am hoping to start injecting it next week. I have also been working on getting videos of the embryos contracting. These videos are not taken under the confocal microscope but are allowing me to observe what is happening to the embryos when they move. I have been trying to get videos of the embryos when they first start making the wave contractions but so far, they have proved to be to delicate. I am happy to report that I have been practicing getting the embryos lined up on the injection dish and have gotten much better at it! This past weekend the OIMB REUs and the main campus REUs all camped together. On Saturday just the OIMB REUs spent the night. Although we were planning to just go tide pooling on Sunday morning, the tides were so low that we ended up going out on Saturday morning too! On Saturday we went to North Cove and saw many different types of algae, lots of worms, and some brittle stars. On Sunday afternoon we met up with the main campus interns and we enjoyed playing Frisbee and talking around the campfire. When we went tide pooling the next morning, we went to South Cove. There was less algae here and more sea anemones and hermit crabs. Dr. Watts even found an octopus! On Tuesday half of the interns gave a presentation on their research proposal. These presentations were more filled out and detailed than the previous presentations that we have given. Four weeks of chewing over what I am doing and will be doing and what I want to accomplish has made it where I can explain my project in a much clearer, concise way. I know that our presentations were all much clearer and easier to understand this time.  Welcome to the week three blog post everyone! For me this week has still been full of learning new and interesting things. I have continued learning about and working with PCR. Polymerase chain reaction is used to make many copies of certain strands of DNA so there is enough DNA to work with. I’ve also been learning how to use the confocal microscope. I was able to get some beautiful and interesting videos of first cleavage with Dr. von Dassow directing me. I also think I’ve got the timing down with the barnacles. Up until this point I usually have had to wait until right before lunch to get a brood so the embryos don’t always have enough time to mature before the end of the day. I also found something that I need to work on. On Friday I tried pipetting some embryos into an injection dish. After watching Dr. von Dassow easily perform this maneuver perfectly I figured that I could at least get the embryos lined up on the dish. Well, I vaporized the first 20 I tried to move and got the next 20 stuck in the pipette. After that I got some embryos that were in one piece on the injection dish. However, they were scattered randomly across the dish. I’m going to make sure to practice before I try that again. We’ve also been busy with events outside of our lab. On Saturday we spent the day on and around the University of Oregon’s main campus. We were able to tour a lab that works with brain activity during movement. Also, since Thursday was the 4th of July, most people had little or no work to do in the lab. In the morning I was able to check crab traps with Renee and Megan. It was very muddy but I enjoyed learning about crabs and their habitats. In the afternoon we had a potluck at Sunset bay. It was a beautiful warm day, perfect for being on the beach. I enjoyed getting to climb down to visit a hidden beach and exploring the many hiking trails that weave through that area. We have our proposal for our projects due on Tuesday so I’m spending plenty of time looking up articles. Putting together a paper has given me the chance to more firmly decide how I will answer my research questions and what I need to do in order to get the final result. We are going camping this weekend with the interns from the main U.O. campus. I am looking forward to getting to know them better and hopefully enjoy a few hikes.  Balanus glandula Balanus glandula Hi everyone! This is week two and I’ve got a project! On Monday Dr. von Dassow, my lab mates, and I discussed what we would be doing for our projects. I will be working with barnacles; Balanus glandula is their scientific name but informally they are called acorn barnacles. They are the most numerous barnacles on the Oregon coast and usually grow on docks and rocks. I will be studying cell polarization in barnacle embryos. In C. elegans cell polarization happens early, during first cleavage. When an egg is fertilized it starts dividing to create new cells. When the egg makes its first division it pushes some types of proteins into one half of the cell and other proteins into the other half of the cell. So, when the cell divides into two cells, one cell has a lot of some proteins and the other cell has a lot of other proteins. This happens because different parts of the creature will be formed from each cell. For example, part of one of the cells will eventually form the gastric tract. This cell will take the proteins that it will need to make this system. The moving around of substances is called cell polarization. I will be trying to discover if cell polarization happens in basically the same way in Balanus glandula as is does in C. elegans. To do that I will be using a probe to track atypical Protein Kinase C. The aPKC is found in many different organisms so there is a good probability that it will be found in Balanus glandula. I will be using a probe to track this protein and see if it will be doing anything interesting in barnacle eggs. Right now, I am practicing injecting substances into barnacle embryos and learning how to use the confocal microscope. This week I attached a video so I could show you what the barnacle embryos look like after a successful injection of RNA. What you will see is a bunch of white lines “flowing” through the embryos. The probe that was injected is a protein that sticks to the front of forming microtubules. When the microtubules stop growing, they either attach to a solid object or catastrophe which means it breaks apart. The video is sped up and is made up of pictures taken at different depths in the embryo. Helping to take this video was my introduction to working with the confocal microscope. If you watch the video closely you can see the cells dividing. On Sunday a group of the Interns went to Bastendorff beach. It was warm enough that we were wearing shorts and t-shirts! I know that I also enjoyed our meeting on Wednesday when we all gave a 5-minute talk on what our projects would be on. It was interesting to learn about everyone’s plans in more detail than just snippets from mealtime discussion.
Hello everyone, my name is Sadie and I’m excited to be able to tell all of you about the knowledge I’m gaining and the research I’m going to do. I’ve always been fascinated by small things that are not easily visible to the human eye so working in Dr. von Dassow’s lab is a perfect fit. I have never had the opportunity to do research, so I am very happy that I get to be a part of this program. While I do love marine biology, I plan to go into the medical field and be a surgeon. That means that everything I learn about cells, techniques, and research will help me a lot down the road. Science has been a big part of my life and I have never doubted that I would choose a career that had something to do with science. I live in Central Oregon and enjoy hiking, reading books and working with animals. I just gradated from Central Oregon Community College and will be attending St Olaf this fall. This week Dr. von Dassow taught my lab partners and I how to use different types of microscopes and tools. Being able to see the structure and the beauty of the plankton and the jellyfish we caught was amazing! While I have been able to look at organisms under a microscope, I’ve never seen anything quite as beautiful as the jellyfish. One of my favorite techniques we did was inserting RNA into sea star eggs. By using an extremely tiny needle and maneuvering it by using a machine we could insert a tiny amount of liquid into the egg. Then we were able to watch the movement of different substances in the eggs. This week the REU Interns also got to go to a potluck at Sunset Bay. While it was foggy, we had a fire going and could warm up between trips to explore the beach. It’s been wonderful meeting and talking with all the REU’s. Since we are all from different areas of the country; I learn a little more about the different parts of the country every time we talk. This Saturday we are going out on a research boat to collect some samples. I’m very excited but a little nervous given the fact that I’ve never been out of sight of land before!
|
AuthorHi! My name is Sadie and I just graduated from Central Oregon Community College in Bend Oregon. I am working in Dr. von Dassow’s lab and I am excited to learn about research and cells. Archives
August 2019
Categories |
||||||||||||
Proudly powered by Weebly







 RSS Feed
RSS Feed