|
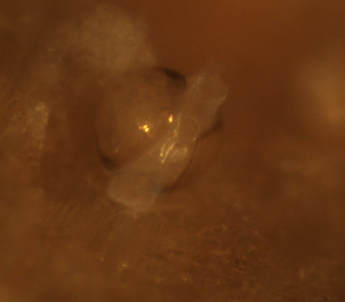
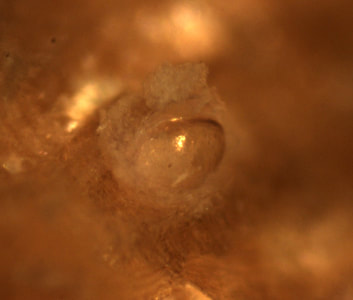
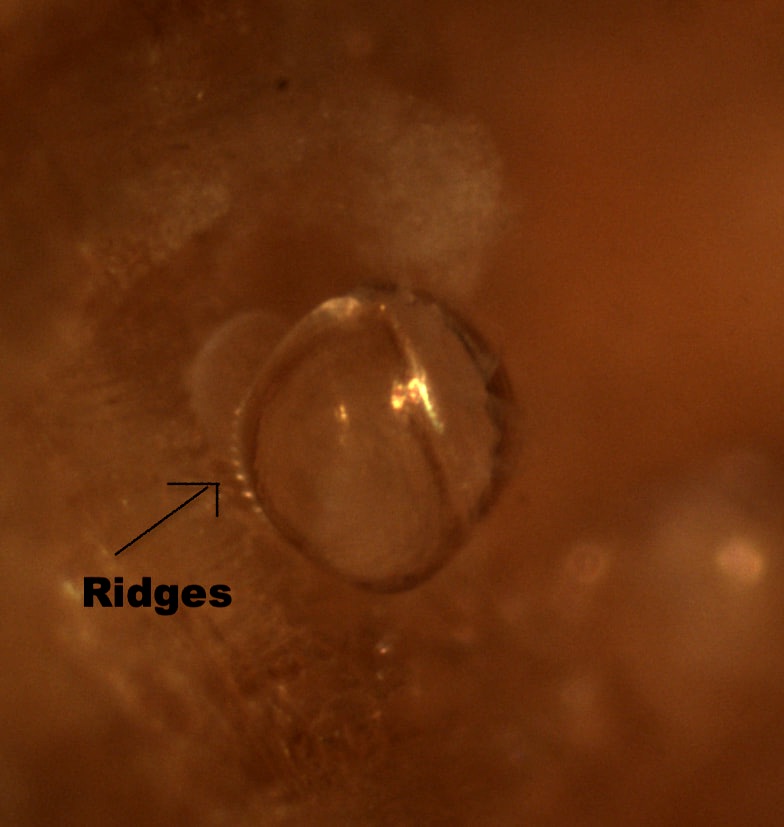
Very exciting developments with T. bartschi this week! After six days of anxiously watching and waiting, I observed my first burrowing behavior seemingly overnight! Ridges along the ventral edge of the valves (shells) had formed, and the larvae were using them to scrape away at the wood. Debris, likely containing mucus and wood particles, was beginning to coat the larvae as they made their way into the wood. After two days of collecting data on burrowing, it was time to begin my second experiment with 36 new samples of wood, which meant ‘out with the old’. I’ll admit, it was a little heart-wrenching watching those little guys go down the drain, but that is part of the process. The next trial will have the same species of wood, same number of replicates, and shipworms from the same collection location. Ridges along the ventral side of the shell have formed  Wednesday, Nancy, Richard, and I spent the morning in a skiff up the South Slough retrieving more of Nancy’s test panels. The tide wasn’t entirely on our side, as a few of the panels required some fancy boat maneuvering by Richard. Nevertheless, we persevered in collecting all 7 panels and replacing them with new ones. On Saturday, our REU group had the opportunity to visit the Oregon State University Hatfield Marine Lab in Newport. Hatfield is a very large facility that partners with six state and federal agencies on vast range of marine research topics. We met with some of their REU students and compared our summer research projects and experiences on two very different campuses. We spent the rest of the afternoon at the Oregon Coast Aquarium before making the drive back down to Charleston. Trip to the Oregon Coast Aquarium
0 Comments
The highlight of my week was the opportunity to accompany OIMB instructor Nancy Treneman during her shipworm field research. Nancy has deployed wood panels in numerous waterways throughout the Coos Bay area and checks them periodically to see if they have been colonized by shipworms. Observing her experimental designs and methods for data collection have been enlightening. For me, this is the type of experience that has made the REU program so rich. Reading the “methods” section of a scientific paper conveys only a snippet of the amount of time, energy, and tenacity it takes to conduct research. There is no room for a story in the methods section. There we were, covered head to toe in mud, creating makeshift boardwalks to navigate the boot-sucking mud. We piqued the curiosity of many passersby, who did slow drive-by inspections and double-takes. It was an adventure. It was research. My experiment hit a bump in the road this week. The discovery of excessive amounts of tannins leaching from my oak samples, which led to mass mortalities in the larvae exposed to that wood, forced me to select an alternate form of hard wood for testing. Also, I committed an egregious error when preparing my wood samples. To prepare them for the larvae, I soak them in seawater for at least 24 hours which helps soften the wood and start the formation of biofilm. In my naiveté, I soaked all of the samples together, in one container. This exposed all 36 wood samples to the harmful oak tannins mentioned above, and contaminated everything I had prepared. Ahhhh, the joys of learning the hard way. Thus, most of this week was spent rebuilding and preparing another trial, which commenced on Thursday. So I finally have larvae in my treatments and now it’s all about watching and waiting for them to settle! Our weekend activity will be a trip to the Oregon State University Hatfield Marine Labs in Newport, with a stop at the Oregon Coast Aquarium. I have been scouting OSU Hatfield as a location for my master’s program, as they conduct extensive oyster research that I would like to learn more about. We will be socializing with the REU students there and I plan to ask lots of questions and take full advantage of this opportunity.
The odd little shipworm is starting to burrow its way into my heart. A few weeks ago I was telling myself “okay, so it’s not an oyster, but the larval stage is close enough so just be happy to study something similar.” Indeed, the shipworm was living in the shadow of the oyster. But nobody puts Teredo in a corner! So here I am, neck deep in shipworm literature (and boots stuck in the mud!), observing them every day and trying to learn as much as I can. They have been stigmatized as destructive pests that turn boats, pilings, and docks into swiss cheese. Their ecosystem services have been eclipsed by the damage they cause to manmade structures, when in fact, the holes that they burrow become refuges for other marine organisms. They cycle carbon just as their relatives the oyster and the clam. They break down wood and remineralize plant material, transforming it into nutrients to be used by other creatures within the ecosystem. Their benefits are vast, so let us not be so hasty in our judgement of the shipworm. Week four has been all about gathering the materials to get this experiment off the ground. The low tide series gave Richard and me two days for shipworm collection. We successfully gathered wood with live adult shipworms from two locations, Shinglehouse Slough and the junction of the Coos River and Isthmus Slough. Cutting the wood blocks and tile, assembling them, then soaking for at least twenty-four hours before experimentation are the next items on my to-do list. On Tuesday, the REU students were asked to submit research proposals to our PI’s and to give 10 minute presentations about those proposals to the entire group. What a wide range of studies we have! From isopod visual capabilities, to the biomechanics of comb jellies, to the prevalence of the invasive European green crab just to mention a few.
Rounding out the week, I had the pleasure of meeting OIMB instructor and shipworm researcher, Nancy Treneman. Nancy has sites throughout the surrounding waterways where she deploys wooden test pallets for shipworm collection. I have been invited to join her next week to retrieve and process the pallets. Her enthusiasm for these bivalves is infectious! I am so thankful for the opportunity to learn from her and work alongside her in the upcoming weeks.  aquarium tanks set up to filter any larva released from shipworms burrowed in the wood aquarium tanks set up to filter any larva released from shipworms burrowed in the wood It has been buckle-down time to get my experiment set up. Three weeks have already gone by leaving only four weeks to collect data and prepare a poster for presentation. With Richard’s help, I set up two tanks fitted with fine mesh filtration containers and filled them with the wood collected from Haynes Inlet last week. If any shipworms release their larvae I should be able to collect it and use it for experimentation. Friday I got my first naturally released pediveliger larvae! I was only able to collects 10 individuals and if I do not get enough larvae naturally I will have to remove them by dissection as I have done previously. I would prefer the natural way though, because the larvae would be at optimal development to begin their free-swimming life stage.  wood square for larvae to (hopefully) burrow into wood square for larvae to (hopefully) burrow into I have also begun preliminary tests with my previously collected larvae to see if they will settle and burrow into small submerged wood squares. The wood has been affixed to pieces of slate to keep them weighted down and placed into small dishes with 20 larvae added to each dish. So far I have observed six larvae using their foot to move around on the wood, but no burrowing behavior. Myself and the rest of the REU interns went camping this weekend at Sunset Bay State Park and were joined by biology REU interns from the U of O main campus in Eugene. We spent the afternoon exploring Sunset Beach and the evening roasting marshmallows and telling stories. Sunday morning, we were treated to a low tide that exposed an extensive intertidal zone at Cape Arago, perfect for tidepooling. When tidepooling in the San Juans, the elusive gumboot chiton, Cryptochiton stelleri, was the most prized discovery for my sister and I. To my delight, C. stelleri was everywhere I looked at Cape Arago! Nicknamed the “wandering meatloaf”, the gumboot chiton is the largest species of chiton in the world. It is a member of the phylum Mollusca (along with bivalves, octopus, and snails), and can grow up to 13 inches and 4 pounds! The REU Crew!
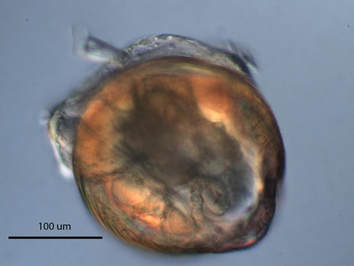
This week I’ve been feeling a lot like the very planktonic larva I have been studying: drifting in a general direction, waiting for the right place to settle. To my dismay, Ostrea lurida, the Olympia oyster, does not seem to be a reliable source of larva at this point in the season. After opening 10 more organisms and finding none containing larva, I had to forfeit the hope of using them as the subject for my summer project. It was back to the drawing board to find a suitable brooding species. What is a brooder? And why must I select one? Brooding refers to the act of incubating one’s fertilized eggs within the body wall until they reach a certain developmental point, then releasing them into the environment. By selecting a brooder, I can acquire a large number of larva from one individual, and if I can identify the species of the adult from which the larva came, I will know exactly what organism I am studying. The alternative would include the extra step of filtering free-swimming larva from the plankton then having to identify that organism. This can be a painstaking process as many larval organisms do not have well-defined characteristics at such an early stage in development. Enter the shipworm, Teredo bartschi. Not the first organism that comes to mind when one thinks of bivalves. The shipworm is not a worm at all, but is named as such due to its worm-like body that is not covered by a shell, as bivalves typically are. Its valves (shells) are found at the anterior tip of the body and have been modified to burrow into submerged wood. Their ability to effectively turn a solid piece of wood into swiss cheese has been well documented. In 1588, the shipworm species Teredo navalis is credited with weakening the wooden ships of the Spanish Armada giving England an advantage that eventually led to the armada’s defeat. My mentor, Richard, and I set out a mission to collect wood samples containing T. bartschi in nearby Haynes Inlet. Thus far, the shipworm has been much more cooperative! Extracting them from the wood is a challenge, as their bodies are extremely delicate, but five of the six I managed to collect contained larva in various stages of development. I am interested in the settlement and metamorphosis of larva, and experimenting with different salinity levels. At last, I have found an organism to use as my research subject. I have settled, and now I can begin my observations. It’s not all work here at OIMB. Last Friday the REU interns, along with my mentor Richard and two graduate students, Nicole and Makenna, went on a dredging trip aboard OIMB’s research vessel R/V Pluteus. Captain Knute even let me steer the boat! Among our haul were numerous sea cucumbers, basket stars, brittle stars, and crab. The sun shone brightly for our entire outing and an afternoon on the water was the perfect way to celebrate the end of a great first week here at OIMB!
|
AuthorHello, my name is Tiffany Spendiff. I am a third-year marine biology student and unrelenting bivalviaphile. This blog is an account of the successes, lessons, trials, and tribulations of a burgeoning marine scientist. Archives
August 2018
Categories |
Proudly powered by Weebly





















 RSS Feed
RSS Feed