 I can’t believe that this was the last week of the internship! The summer went by very quickly and I still have so much that I want to do! This week I continued to take videos and pictures of the embryos. Since I had collected all the data I needed, I was able to try some different methods that could possibly make the process easier. We tried to soften the egg shell to see if it made the eggs easier to inject and easier to put on a slide. The eggs did remain easier to inject for a longer period of time, but they were very delicate and didn’t enjoy getting stuck with a needle. Everything on the inside of the cell tended to pour out when the egg got poked with a needle. However, the eggs were considerably easier to flatten on a slide. Usually the egg shell is so firm that it gets broken before it gets compressed. On Thursday I tried injecting Nina’s probe into some barnacle eggs. Since it lights up so well and clearly showed part of the cell in starfish embryos, we though that it was worth a try in barnacle embryos. While the probe was expressed (It light up) in the barnacle embryo it didn’t attach specifically to anything in the embryo. So, it was there but it didn’t show us anything useful. On Friday we had the poster session. I enjoyed getting to share what I had learned with all the people who walked through. A condensed version of my poster is that my research is on barnacle embryos and how cell polarization effects the cleavage furrow pattern. I injected different RNA or protein probes into the embryos and then used a confocal microscope to track the probes in the cell. I discovered that barnacle embryos have an asymmetrical first division that results in one large and one small cell. This division is related to cell polarization. Before first division the cell’s contents are polarized into granular yolk and globular yolk. After the cell divides there is a small cell that contains mostly granular yolk and a large cell that contains most of the globular yolk and a little of the granular yolk. The way that the barnacle embryo accomplishes this is by using two cleavage furrows. The primary furrow forms down the center of the embryo and then a second medial furrow forms and meets the primary furrow in the middle. The cell continues to have this polarization in the later stages. The small cell divides into approximately equal daughter cells but the large cell divides into a large cell that contains mostly globular yolk and a smaller cell that contains mostly granular yolk. The result is three approximately equal small cells and one large cell. I have really enjoyed this internship. It was incredible to be able to learn how to do research and then to work on a project. The skills that I learned are ones that I know will be able to apply later in my career. The professional development sessions have given me more knowledge to use as I continue with my education. Thank you, Dr. Watts, Dr. Emlet, Nicole, and Dr. von Dassow for this incredible opportunity. I also enjoyed getting to spend time with all the other interns and I really enjoyed getting to work with Philip and Nina in the lab. Thanks for the awesome summer everyone!
0 Comments
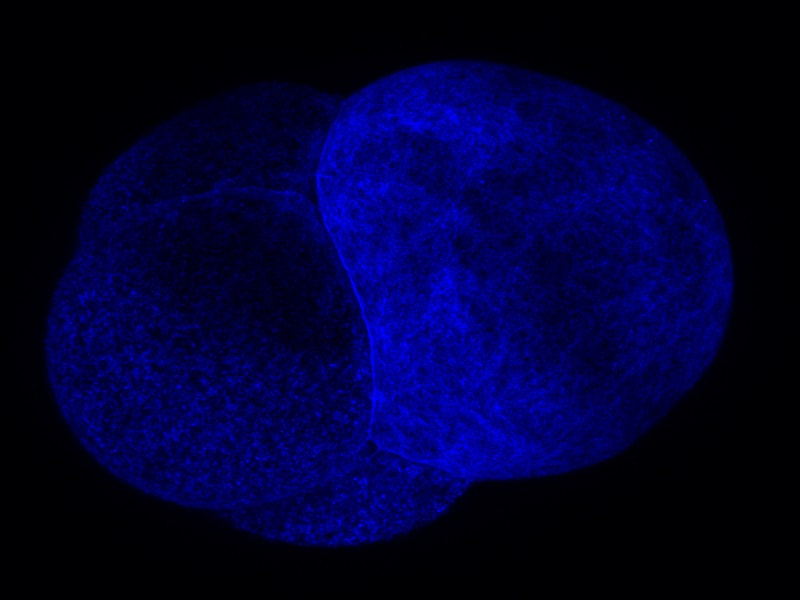
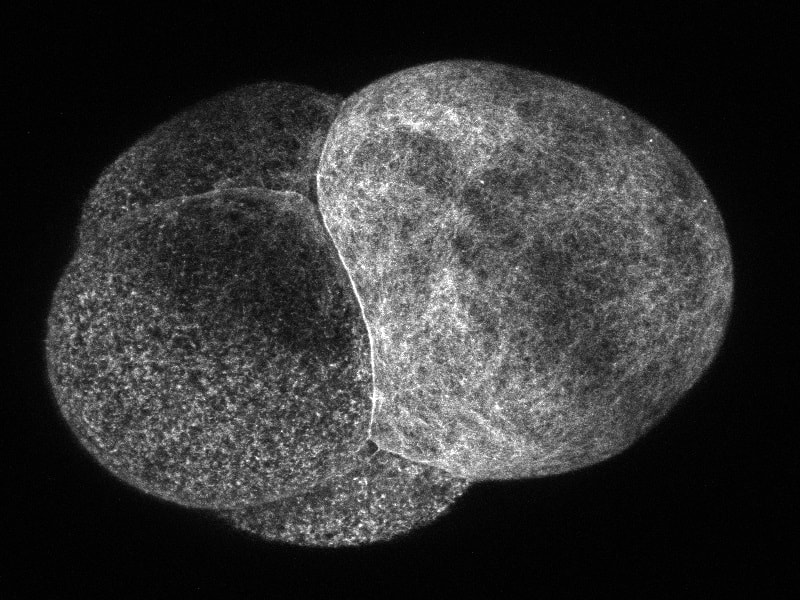
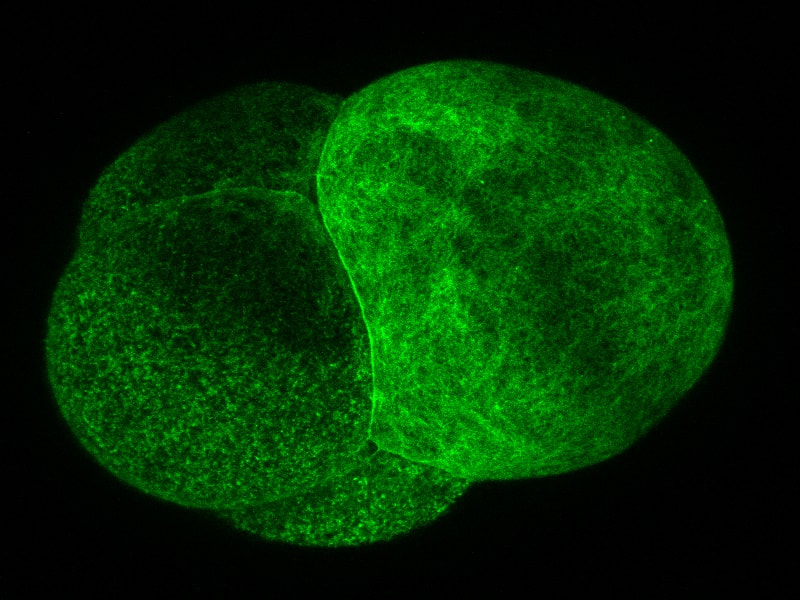
Welcome to the week 8 post everyone! This week we’ve been doing a lot of work on our posters. On Tuesday we had a poster critique among the interns and Dr. Watts. We projected everyone’s posters up on the screen one at a time and gave feedback on what we liked, or thought could be changed. I greatly appreciated the advice. It was fascinating to see everyone’s posters taking shape. Then we turned the posters in for the final review on Friday. It has been surprisingly difficult to cram everything I have seen and observed onto one poster. The most constant comment I got at the critique was to get rid of a lot of the text! Trying to condense everything has made me sift through what I have observed and try to pick out the most important things. Probably the most important finding that we observed was a secondary cleavage furrow. The first and main cleavage furrow starts out like it is going to divide the embryo into two equally sized cells. Then a second furrow forms on the side and continues moving towards the center until it connects to the main cleavage furrow. The result is that there is one large cell and one small cell. The other thing I’ve been doing is sorting through videos and pictures. Dr. von Dassow taught me how to use some basic functions with ImageJ Fiji. The program is incredible, and I’ve had fun turning different probes into different colors to emphasize different pieces of the embryo. Normally we work with grayscale images because that is the best color range to pick out details in. Greens also work well but blue doesn’t show details at all. And while some color combinations, like green and magenta, clash they actually show off the different parts to the maximum advantage. I’m going to attach some images so everyone can see the difference. While I have enjoyed getting to look through the data and explore ImageJ I’m always glad when I have time to inject and image more embryos. Since I have all the data, I need I have been able to some new ways of setting things up and see what else I can learn with out the pressure of getting a result. On Friday we tried putting the embryos in a chemical that keeps their egg shell soft and pliable. Unfortunately it is a lot harder to get the embryos to stick to the injection plate while using 3 APA but we’re still working on it. Below you can see the pictures of the embryos, they are all the same brightness but you can see the details on the cell surface much better in the gray than the blue. Welcome to the week 7 blog post everyone! This week Dr. von Dassow was gone for a conference and Monday was a bit of a struggle. I quickly found that I can’t take 15 minutes to get a needle loaded if I want to get a plate of embryos injected before they get too hard! However, by Tuesday I had figured out how to time everything. I also got a lot faster at doing everything which helped a lot! I spent this week collecting data and trying to learn everything I can by watching the embryos. This Thursday yielded some exciting results for me. I think I’ve found a way to successfully predict where the smaller cell will divide from. Before the embryo divides a layer of something containing the utrophin probe sweeps across the surface of the embryo. What I’ve found from taking and watching videos is that the last spot to be covered by this “hood” will most likely be directly opposite where the smaller cell will be. So, by using a utrophin probe and an EB1 probe that tracks the tips of microtubules, I was able to get a couple of videos showing the cleavage furrow in one day. So instead of getting 10 videos with only 1 or 2 showing the cleavage furrow, I got 2 out of 2 videos showing the cleavage furrow. I have attached the first video that I got on Thursday below so you can look at it. I was extremely excited when I was watching it! If I’m right about the importance of the hood than it will be much easier to find embryos in the correct orientation to film. However, right now the only way I can see the hood and tell which way it is moving is by taking about 3 minutes of video. This works well if the second or third embryo I look at is oriented the right way, but I only have a certain amount of time between when the hood starts forming and when the cell divides. So, I only have time to check a few embryos before they start dividing. I’ll work on testing this idea and hopefully I won’t be getting any more videos that are looking at the back of a dividing embryo! On Wednesday I got to watch two interesting presentations. The first was a defense of a marine biology honors thesis by Alexander Hethman. I’ve never watched a defense of a project before and the topic was interesting. Alexander knew a lot his topic and I really enjoyed getting to hear his defense. We also had Dr. James McClintock speak for our seminar. Dr. McClintock has been doing research in Antarctica and told us about the effects of climate change on Antarctica. He has personally observed changes in Antarctica and showed diagrams of the receding ice sheets and glaciers. He also had some funny stories to tell about his time in Antarctica and as he spoke to others about climate change. On Saturday we will be spending a few hours in the CMLC tell the people who visit the museum about our projects. It should be a lot of fun.
|
AuthorHi! My name is Sadie and I just graduated from Central Oregon Community College in Bend Oregon. I am working in Dr. von Dassow’s lab and I am excited to learn about research and cells. Archives
August 2019
Categories |
||||||
Proudly powered by Weebly


 RSS Feed
RSS Feed