|
This week was extremely fast paced and invigorating. Loie and I got started on TEM sized slicing, made probably around 20 semi-thin slides of both Ifremeria and Alvin, and took countless photos of our slides. I think we are both enjoying each others company in the lab because we can problem solve and trouble shoot questions out loud to each other, pushing our work to a higher quality. I can say I feel much more confident in my work this week than I have previously. While all of this is great news, we are experiencing a few road bumps that will take some extreme digging to solve and rectify. First for my project, I need to look at the tissues collected and determine if they are gonad or brood pouch. This sounds simple enough until I realized that this species’ reproductive ultrastructure has quite literally never been looked at. There is one piece of literature co-authored by Craig that includes some photos of the brood pouch, but not in the histological format that I am using. So, we are having to identify structures using the compound light microscope to determine what we are looking at. This has called for major digging on the internet for any information about the reproductive systems of similar species, but as you can guess, there is almost no literature on them as well. Oh, the beauty of working in deep sea! I did have a breakthrough on Friday though, where after about two days of brainstorming, reading old literature, comparing scattered bits of information, I found what we determined to be larva in a brood pouch! I had a hunch that we were looking at something special and now I feel like a real morphologist. Luckily, problem solving like this is something I really enjoy. Now that we have this bit of information, we can hopefully better identify other tissue samples and work towards labeling key structures for our presentation. Our next bump in the road is that our ultra-microtome is not slicing down to TEM thickness (60nm) properly. The slices we are getting are inconsistent and unreliable. When working with a TEM, the measurements must be precise or else the micrograph photos will not take. We have been trouble shooting all week, messing with the various settings and even setting up an anti-vibration desk, but nothing has worked. Basically, this puts my entire project at risk because we are unable to see bacteria at anything larger than the TEM size. Last minute, Craig had the idea of traveling up to Eugene for multiple days in order to use their ultra-microtome and then use the TEM there as planned. We have an email out to their team to ask if there is availability, so fingers crossed! As of today, we have 11 days to finish our project, make our poster, and fly to Brazil. Not to mention I also have to make my poster for the REU symposium happening the week after we get back. In all honesty, I have a feeling that we will be able to handle this and it will all work out. Maybe that’s my inner unrealistic optimism, but whatever the case, everything will happen as it should. The lab has put our heads together and overcome a lot of obstacles so far, so I am very confident in our abilities. Also, yes I am still have lots of fun in my free time with my cohort! We took a last minute trip to Eugene on Friday and stayed until Saturday night, which was a lot of fun. Eugene is a great little town with some very yummy food. I’ll just leave photos that can do the talking. Thanks for following along!
1 Comment
Before I begin talking about the ultra-scientific things we did this week, I have to share this bit of news that has been successfully distracting me for the last few days: I will be presenting a poster at the International Symposium on Chemosynthetic-Based Ecosystems in São Paulo, Brazil a little under 3 weeks!!! The stars aligned super last minute and Craig offered the opportunity to me and another undergraduate at OIMB, named Loie. We will be presenting on the reproductive ultrastructure of a few species of hydrothermal vent snails. If you know me, you know that I love chemosynthetic life and will trap you in an uncomfortably long conversation about it (whether you like it or not), so being able to meet other people who feel the same way is just perfect! It’s going to be a busy next few weeks, but I feel ready to take on the challenge. (I am freaking out) I also owe Friday of week three a shoutout. I accompanied the deep sea class at OIMB on another Megalopa day where we had the opportunity to take out the ROV. The students (and myself) even got to drive the ROV! It was perfect until we lost the ROV (oops) but quickly recovered it about 20 minutes later. Craig says it’s good experience to lose oceanographic equipment and find it, as it’s not uncommon on research cruises. Overall a success! Quick methods lesson! Week four was all about getting my project officially underway. In order to slice the reproductive ultrastructure tissues with the ultramicrotome, we need to dehydrate them with ethanol and infiltrate them with spurrs resin, making it so the samples will stay intact while slicing. We start by selecting 5 Ifremeria tissues from the lab fridge and assigning them individual ID numbers. After that, we dehydrate the tissues in a series of ethanol concentrations, which pulls all the water out of the tissues (spurr and water do not mix well). To create the spurrs resin, we need to mix together four pretty intense chemicals under a fume hood and with protective gear on. I feel like such a scientist when I do this process. After the spurrs resin has been prepared, we place the dehydrated tissues into increasing concentration mixtures of spurrs and ethanol over a series of 24 hours. Once the infiltration is complete, we can begin placing the tissues into individual molds for the setting process, which takes place in the lab oven. We set the temperature to around 70-80 C and leave them for 11 and a half hours, so the resin can harden thoroughly. Without this process the tissues would simply crumble apart as we sliced them, so it’s very important. We successfully prepared my first round of resin blocks and now I can begin slicing them for viewing. Once we determine which samples are the most promising for producing solid TEM results, we mark the block and begin slicing them at TEM thickness in preparation for our trip to Eugene. Loie will be returning to the lab on Monday so I am looking forward to having an experienced undergrad working with us as we move forward, as she has worked with TEM sized slicing in the past. As for now, before the beautifully hectic next few weeks begin, I will be seeing the Barbie movie with the other REU’s and thrifting to our heart's content. With so much going on, I find it hard sometimes to recall all of the awesome other things that I do outside of work. Being able to socialize and relax with my cohort is really important to me and we’ve managed to have a lot of fun. I’ll include some photos that can speak for themselves :) thanks for reading, ciao!
The week started off beautifully chaotic with a camping trip to sunset beach with the all of the REU’s. We got in Friday night and camped through Sunday morning. On Saturday, we went tide-pooling, where we found so many cool invertebrates like kelp crabs, chitons, purple & red sea urchins, and more. We had the best time nerding out together over our one true shared interest. Later that day, we hiked for upwards of 4 hours along the southern Oregon coast and shared in each others exhaustion on the way back. That night, El showed off her amazing story telling skills with a spooky tale about some campers, and Ethan shared a drawn-out joke about mountain elves, which cracked us up too much. We cooked over the fire for every meal and Maya was awesome enough to bring us coffee from OIMB each morning, which was just the best! I’ll let my photos do the rest of the talking, because this week has taken up the remainder of my head space (in the best way). Progress with my project is going smoothly, but I had some off days on Wednesday and Thursday. Working with such tiny tissues and precise equipment can be taxing on your brain after a few days, and I guess I’m finally feeling that! I started to not be able to get clear slices and kept making little mistakes while staining that would impact an entire slide of slices. Working with Ifremeria in this capacity is still relatively new for the lab, so a lot of our methods are still trial and error, but that doesn’t make me feel much better when I’m knee deep in the process. This week, we had a speaker come for professional development and talk about applying for jobs and working towards your career, which included a part about understanding and acknowledging your strengths and weaknesses. This had me really thinking about what I consider to be my biggest weakness, which is that I often take my mistakes to heart. I feel as if my mistakes, in an academic or research environment, are a much bigger deal than they actually are and define who I am as a scientist and student. It also makes me feel undeserving to be in the position that I’m in today (aka imposter syndrome). In reality, I know that isn’t even close to the truth, but it’s sometimes hard to remember in the moment. That’s something I hope to work to work on this summer, and with such a kind, intelligent, and patient team in my deep sea lab, I think that it’s possible. (Update: Turns out I had the wrong settings for the ultramicrotome and was able to get a perfect sample by the end of the day Wednesday!!) Next week, we have a group of REU students from Oregon State University coming to visit our labs and get to listen to our individual projects. Our REU cohort will be leading the tour, that can be pretty nerve wracking, but I’m excited! At UVI, I often gave tours to visiting researchers at our mangrove restoration lab and it’s something that I really enjoyed. I can’t wait to share my project with a new group of people and listen to their interests, as well. I will also begin baking my reproductive ultrastructure tissues in resin on Monday, which will officially mark the beginning of my research project. This will begin the process of collecting the slices and eventually taking them to Eugene where the TEM is. I’m looking forward to hitting the ground running next week and can’t wait to share my experiences with that. Thanks for reading, à bientôt!
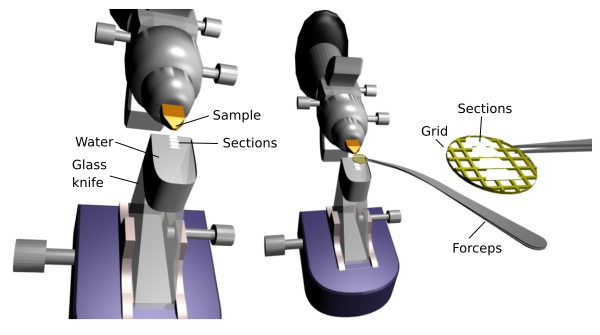
P.S. Aquatic Nocturne is a poem by Sylvia Plath that has been on my mind this week, here it is. Week two has been pivotal in my overall confidence in my abilities as a scientist, and as a person. The week started with the cohort able to spend time together over the weekend kayaking and paddle boarding at a nearby lake, tumbling down sand dunes, hiking overgrown deer trails, and solving unnecessarily difficult puzzles. At first, I was secretly nervous to meet the people I’d be spending the better part of the next two months with, but its become increasingly clear that I have nothing to worry about. Everyone I’ve met is so authentic and open, I feel like I’m going to learn so much from them and be able to find new parts of myself that I’ve maybe lost with age. Already, I think I could identify a good amount of common birds and be able to tell you all about carnivorous larvae (you know who you are). Being able to come out of my shell and immerse myself in a new environment has always been tricky for me, but I’ve been shown time and time again that it pays off in the end. This week at the lab, I’ve been focusing on familiarizing myself with the machinery I’ll be using for my research project. I’ll be looking for the presence of symbiotic bacteria in the gonads and brooding pouch, referred to as the reproductive ultrastructure, which will allow us to determine the developmental stage that they receive the bacterium. Quick Methods Lesson! The senior lab members have already collected the snails and extracted the necessary tissue samples from the snails (thank you Lauren and Caitlin!). So, I’ll begin by placing the tissue in resin, slicing the resin into 60nm thick pieces using the ultra-microtome, placing the slices onto grids, and transporting them to the main campus in Eugene where the T.E.M. (Transition Electron Microscope) is located. With the T.E.M., I’ll be able to visually observe whether or not there is a presence of symbiotic bacterium in the gonad or brooding pouch. Each step requires precision and lots of practice, so I have wasted no time getting a head start on becoming comfortable with the process. Along with that, on Wednesday the interns gave our five-minute talks where we briefly presented our project goals and methods, which was a great opportunity to organize my thoughts and practice discussing my research in a cohesive way. Thursday might have been one of my favorite days so far. My mentor Craig invited me and my lab partner David to tag along with him on a trip up to Newport to grab a part for our ROV, where we saw the most stunning scenery, listened to old natural history tales, and talked about our traveling adventures. I’ve decided that I’m gonna have to do that trip again sometime. With being in a deep sea lab, I’m often asked how I got interested in the deep sea. This question stumps me, because I haven’t really found a great way to explain. Growing up, my dad and I spent our nights watching “How the Universe Works” with Morgan Freeman, where I first got a taste for the wonder that is the unknown. Each episode felt like a cliffhanger, leaving me only wanting to know more. As I got older, I heard the commonly repeated fact that only about 5-10% of our oceans had been explored, and the deep sea was a relatively untouched mystery to scientists. This sparked the desire of my childhood of unveiling such mysteries, challenging me to explore the darkness and bring the unique beauty of it to the surface. In my mind, the deep sea below and the universe above are like reflections in a mirror, an “as above so below” type of situation. I believe a lot of secrets to the universe and the origin of life on Earth are hiding in the deep. Wouldn’t that be cool? I think that’s it for this week, over and out!
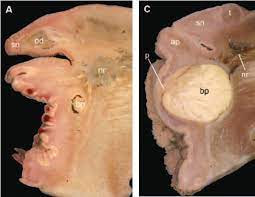
My name is Tara and I’m a marine biology student at the University of the Virgin Islands. originally from the beautiful, land-locked state of Oklahoma, where the ocean is a five hour drive away. Though I only visited the ocean a handful of times in my childhood, I never had a doubt in my mind that I was going to spend my life studying the beauty and mystery of the marine world. I made the decision to move to the Virgin Islands in 2021 to begin my marine biology studies, where I have had the most incredible experiences surrounded by inspiring people and the deep blue sea. On St. Thomas, I spend my time snorkeling, scuba diving, hiking, and working in a mangrove restoration lab. These experiences have allowed me to cultivate my love for marine science and explore the different niches available for research, including deep-sea hydrothermal vents. This led me to OIMB, where I will have the opportunity to study these ecosystems and lives of the organisms that live there. This summer, I’m working with Dr. Craig Young, researching deep-sea hydrothermal vent invertebrates. Conditions at hydrothermal vents are harsh and unforgiving, but the organisms adapted to thrive in these conditions are as fascinating as they are peculiar. Through partnerships with chemosynthetic symbionts, vent species are able to meet their metabolic needs in environments that receive no sunlight. Each species found at vents is idiosyncratic and specialized. For example, Ifremeria nautili, a species of Gastropoda, has unique adaptations for reproduction and dispersal. The females have a unique brood pouch on their foot that is a modified pedal gland. A recent discovery made at OIMB revealed that Ifremeria reproduce with a a novel larval form, named Waren’s larva, that is not found in any other species.
I will be working with Ifremeria nautili this summer, in hopes of contributing to the research that will determine at which developmental stage they obtain their chemosynthetic symbionts. My internship will involve lots of lab work with the electron microscope, but I’ll also have the opportunity to participate in deep sea trawling boat trips on the new OIMB vessel, the Megalopa. I already sailed on my first trip on Wednesday, which was such a cool experience (minus the seasickness). We trawled as deep as 500ft and found some incredible creatures. I’m also starting to familiarize myself with some pretty high-tech equipment in my lab in preparation for the work I’ll conduct later in the summer. This week has shown me that this summer is going to be very eventful. I feel so lucky to be able to study my favorite ecosystem and be surrounded by such a great team of interns! |
AuthorHi, I’m Tara and I’m a marine biology student at the University of the Virgin Islands. This summer, I will be studying under Dr. Craig Young on a deep sea species of snail. I will be focusing on the reproductive ultrastructure of Ifremeria nautilei . I’m particularly interested in deep sea hydrothermal vent invertebrates and other life in extreme environments. Recently, research on these environments has been used in the search for extraterrestrial life in our solar system. (How cool is that?!) When I’m not in class, I am working at my university’s mangrove restoration lab as an intern, or assisting in the science 100 class as a peer instructor. Outside of academics, you can find me at the beach with a book in my hands surrounded by my friends. ArchivesCategories |






































 RSS Feed
RSS Feed