|
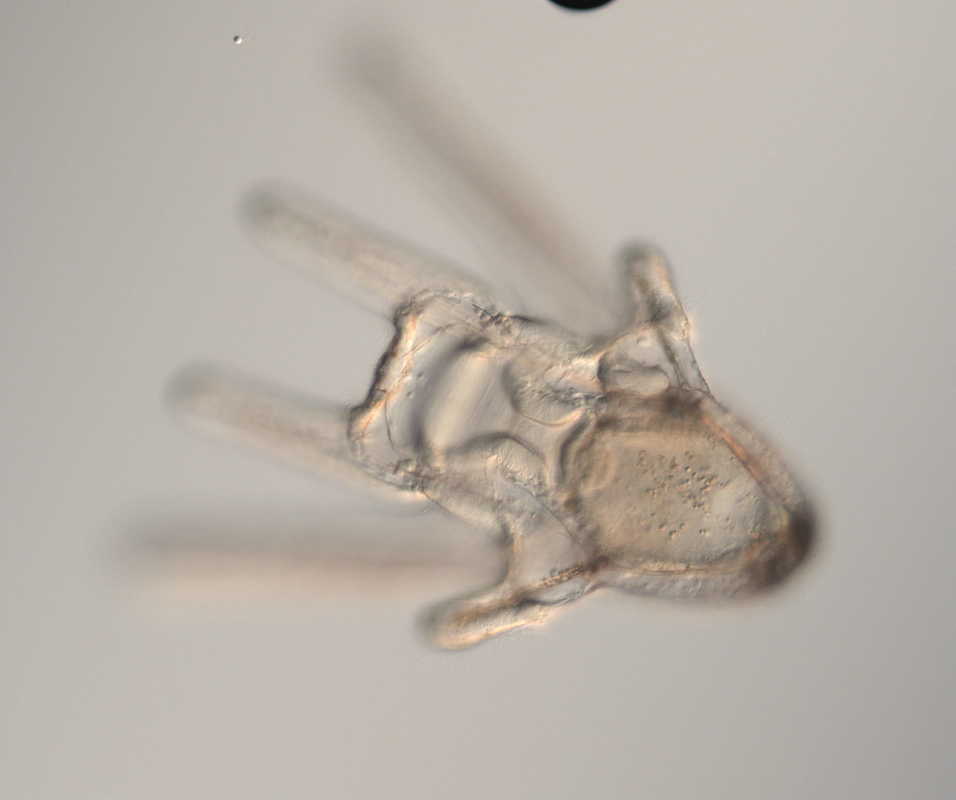
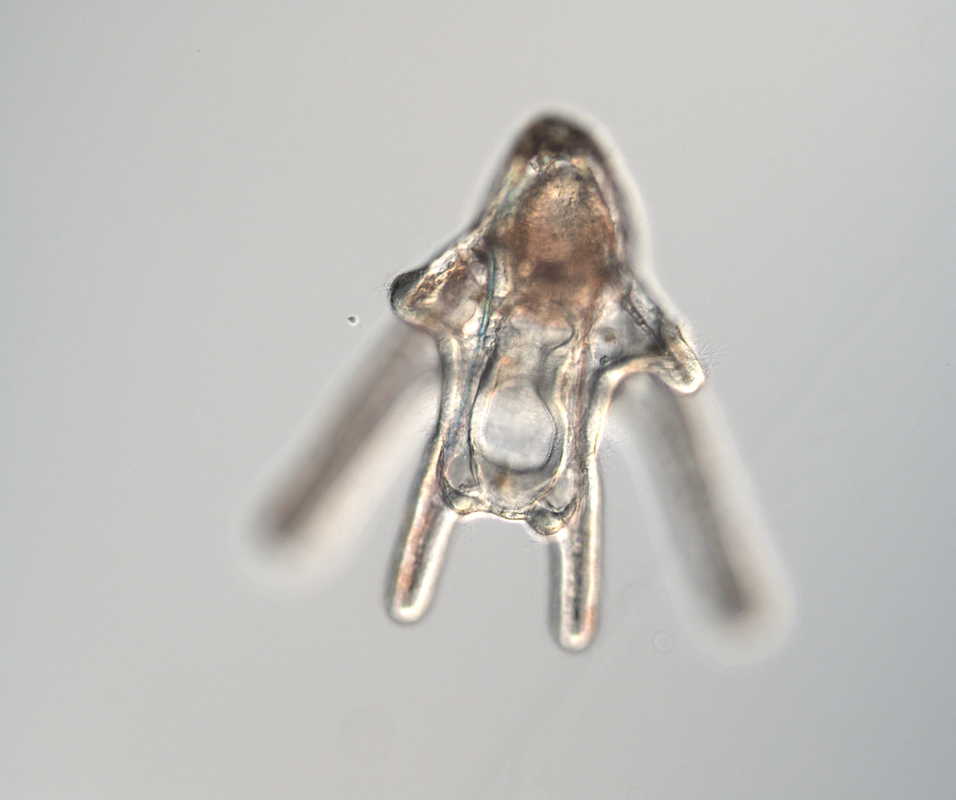
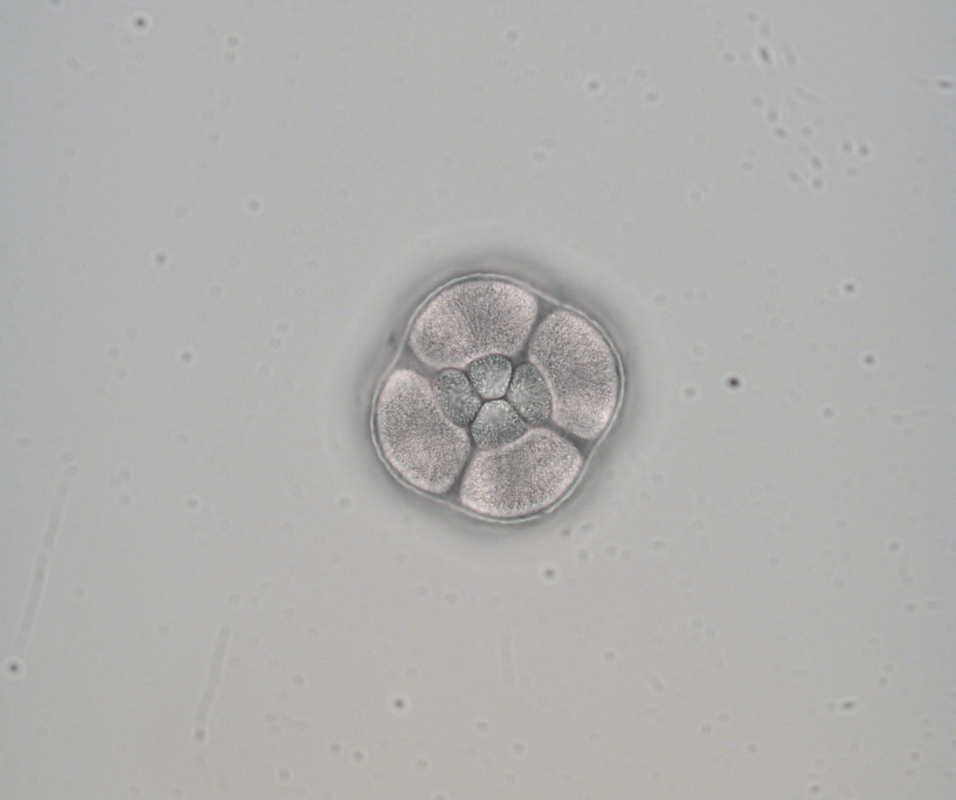
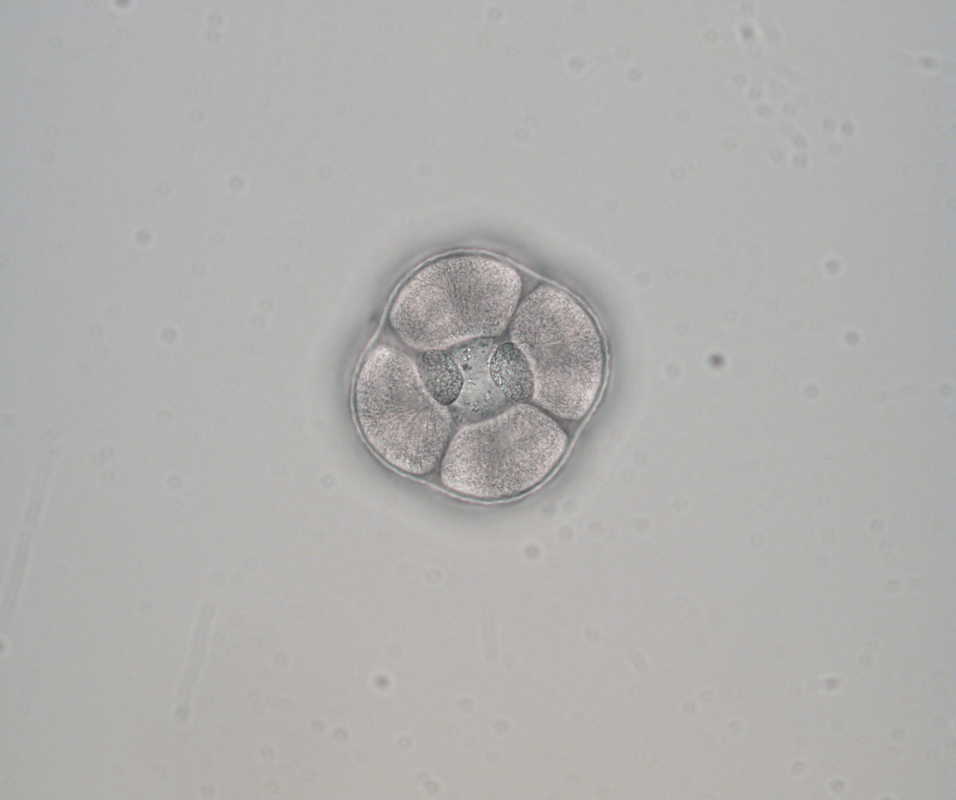
Hello and welcome back! What I’ve learned this week is that there’s really no better way to start your morning than by shaking some urchins. Seriously, try it some time. I know it seems counterintuitive to get a good grip on a spiky ball in the palm of your hand and to shake it around forcefully, but it’s actually quite a magical trick. The most common way to spawn urchins is with an injection of potassium chloride (KCl) – this stimulates the gonad wall to contract and, if you’re lucky, lots of sperm or eggs will be released. A female can release millions of eggs during each spawning, and this is way more than we’re actually able to use in one experiment. When urchins are vigorously shaken, however, they only release a portion of their gametes, allowing us to use a given animal again for future spawning. Once they’re done, we put them back in the tank and let them relax for a while. As I gradually become more comfortable in the lab, time seems to go by more and more quickly. Last week, we started growing cultures of whole, half-, and quarter-sized sand dollar larvae in order to better understand how ciliary band and mouth proportions scale with changes in size. In order to create partial larvae, we must dissociate embryos at the 2- and 4-cell stages to form half- and quarter-sized larvae respectively. In addition, we injected one batch of embryos with GFP-centrin, allowing us to visualize ciliary band density using confocal microscopy. We have been like paparazzi or first-time moms with our little larvae, taking loads of pictures of them as they grow up. Left: Whole-sized sand dollar larva at 9 days old (20x magnification) Right: Half-sized sand dollar larva at 9 days old (20x magnification) In addition to creating partial sand dollar larvae, we began manipulations with purple sea urchin (Strongylocentrotus purpuratus) embryos. At the 16-cell stage, urchin embryos have 4 small cells called micromeres. These form on one side of the embryo and are responsible for development of the skeletal rods. Sounds pretty essential, right? Well, when these cells are removed, the embryo is not necessarily doomed – other cells around it can compensate and help to form skeletal rods. Alas, the larva survives… but not without some consequences. Micromere-less urchin embryos give rise to plutei with all the right parts but with altered proportions and undersized mouths. With these tiny-mouthed larvae, we can investigate the impacts of this manipulation on prey capture. The way we do this manipulation is, might I say, super cool. We use LASERS! We shoot embryos at the 16-cell stage with what is essentially a microscopic machine gun, taking out their micromeres and watching them explode under the microscope. It’s a great way to get out some frustration after a long day. Left: A wonderfully intact purple urchin embryo at the 16-cell stage Center: Two micromeres blasted, two more to go Right: A micromere-less purple urchin embryo Research alone has been a lot of fun, but we have had the opportunity to do a lot outside of the lab as well. Last weekend, we all went camping at Cape Arago along with students from the REU program at the main campus in Eugene. Some intense s’more making happened, and one REU may or may not have almost set his pants on fire with a flaming projectile marshmallow. On Sunday morning, we went tidepooling and I was blown away by the amazing biodiversity in the Oregon intertidal. We got to see urchin beds, nudibranchs, sea stars, anemones, sea cucumbers, crabs, and more. I have to say, tidepooling in the Pacific Northwest is really, really spectacular (sorry East Coast!). On the fourth of July, we attended the OIMB fourth of July picnic and participated in the annual egg toss. Juan and I made it to the top four thanks to a very, very tough egg that was willing to take a few falls. That night, we got to enjoy an amazing sunset and watched fireworks from the beach by the OIMB campus. I’ll say it every week, but time here is truly flying by and I am trying to cherish every moment! Thanks for reading!
0 Comments
Leave a Reply. |
AuthorHello! My name is Ana and I am a rising senior studying biology and music at the College of the Holy Cross in Worcester, Massachusetts. This summer, I am working under the mentorship of George von Dassow. I am looking forward to seeing where my research takes me and to becoming a part of the OIMB community! Archives
August 2018
Categories |
Proudly powered by Weebly








 RSS Feed
RSS Feed