|
It’s the halfway point of our summer, and we all find ourselves a little flabbergasted at it. How quickly the weeks go by when you love where you are and what you’re doing! Whenever someone brings it up at the table, we make sure to answer them with an enthusiastic collective “Noooo!” This week, with our methods and trajectories settled out, we presented our project plans to our program heads and each other. I loved hearing about what my friends spend their days doing, and how widely varied our projects get, from genetic sequencing at the bench to crab-wrangling on the marsh. 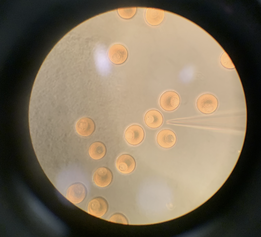 I also started a new test on my Patiria eggs: injection with MPGAP (aka RGA3/4, aka ARHGAP11a, don’t you just love cell biology nomenclature?), a molecule that tamps down Rho activity and dampens the waves on the cell surface. We’ve tried turning our amplifier up, now we’ll try turning it down too. Which speakers will crackle with feedback? A cursory review of our data, however, has also revealed plenty of opportunity for troubleshooting. The eggs in our setup seem to have a harder time with division than we expected. Perhaps it’s the season of the year affecting the starfish reproductive cycle, or perhaps it’s because the cells get squished under their slides. Or maybe our fluorescent probe, a glowing protein fragment that we use to light up Rho under the confocal microscope, is giving them trouble. We’ve started adjusting our protocol to get to the bottom of it.. On the subject of getting to the bottom of things, the REUs did something rather awesome this last Saturday. With pants tucked into boots and ginger snacks in our bags, we set sail on the Research Vessel Megalopa for a dredging expedition. From the jetties of the harbor, fogged as always, we followed the seals and common murres out past the edge of the bay, and there we winched down a grab bag to scoop up a sample of seafloor critters. It was a perfect day for dredging, calm on the water so we could crowd around the sorting table like kids at a candy store. 300 feet down, the world grows dim; detritus-feeders and small carnivores reign. We met familiar animals and bizarre ones: neon-red soft corals, twisty basket stars, scaly worms. I can’t imagine what these fellows were thinking, if invertebrates can be said to think. Most of them had probably never seen full sunlight before. It’s amazing to be surrounded with people who all take as much interest as we do in this strange squishy marine world. And it’s easy to forget that our projects take us to far-apart corners of the estuary and the field of biology - when we talk to each other about research, it feels like we’re all on the same path, encountering the same delights and mishaps. That’s science for you!
0 Comments
My research felt a little like that trail this week. Really it’s been as much obstacle as pathway, and only repetition can clear it. But I know I'm getting somewhere. With methods like ours, you can spend a full day on preparation, and only realize the next day that you bungled it severely. I might mishandle the eggs, or pick a bad batch, and not know until it’s too late. Tuesday was particularly vexing, since we switched over to a new batch of RNA. The cell-city is an exhaustive bureaucracy, and this project puts me in the forgery business. The RNA we deliver must be signed in polyadenlyated triplicate, stamped with 7-methylguanosine, et cetera. All kinds of minutiae can change its effectiveness. Our new batch came out much more potent than the stuff we’d been working with before, and on top of that, something seemed fishy about the eggs too. After getting careless with the timing and then rushing to arrange everybody for the camera in proper order, I watched my precious cells rally for division, only to scrunch themselves into the most horrifying popcorn shapes. But we make what we can of it. Every day I still get more comfortable with our tools and familiar with the laws of the microverse, and I begin to accumulate the almighty data. And hey, now we know the proper dosage for our new RNA: On Friday we tried again with our calibrated doses, and got a pretty good run. In lab, not every unexpected development is an unwelcome one. This week I caught a firsthand glimpse of larval metamorphosis as one of Chloe’s subjects, confused, attempted to settle and transform in her dish, pushing its digestive tract out through its side. Another day, while I was injecting and Chloe was filming, George brought his guitar into the scope room, and we learned a sea shanty.
There ought to be a sea shanty written from the perspective of a marine larva. The narrator rides the current, braving storms and monsters; 99% of its original crewmates perish, and when it reaches port at last, it breathes a sigh of relief, turns itself inside out, and becomes a worm. A noble life indeed! This week began in fire - a campfire. We spent Friday evening through Sunday morning at Sunset Bay, trekking the shoreline all day and huddling around the firepit, like hydrothermal vent creatures, late into the night. There Ethan exposed us to the horrors of the highly elaborate pun, Shreyaan had a birthday, and Naia, Audrey and Chloe inaugurated a minor deity (a bag of jalapeño kettle chips). While camping, we once again visited the intertidal, this time at a straight channel worn into the rock, a geology like the splitting edge of an old book's leather spine. Riley, a TA at OIMB and one of our trip leaders, taught me the names of seven or so seaweeds, from iridescent Mazzaella to lumpy Leathesia. We cracked open a sea urchin and scraped out the yellow meat from its hull: uni, the animal’s gonads. Now I’ve eaten invertebrate eggs while studying invertebrate eggs, and I can say with all honesty that they were delicious. By high tide that day we were up above the headland’s cliffs, finding our way from cove to cove through tracts of woodland and meadow. The weather turned bright, just in time, and we could see out to the bar where seals lay watching us. Dandelions and rockweed all day… then back to our fire for the night.
When we watch the cells divide, we’ll have to do so with multiple techniques. Some of our properties of interest can be measured in bulk, from footage under normal light, but some will require close-up images from the confocal microscope, using its laser to visualize dyed features of our cells. This all involves a lot of sitting in side rooms, peering into an eyepiece, with one hand on a precision tool and the other on the microscope controls as I fiddle around with egg cells. I’m sure plenty of people would find this the height of tedium, but I’m sincerely enjoying myself. It reminds me of the middle phase of a drawing, when I trace over my pencil sketch with linework, repeating the details until everything’s complete. As I get used to the tools, I’ve been able to watch myself improve at my procedures. Last week, egg sorting frustrated me, but now I can use the micropipette to pick up individual eggs! And I only kill (with freakish consistency) 11% of the eggs we subject to the microinjector. Under the confocal and its singing tunable crystal - yes, really - the cells glow and squirm as they multiply, doing the dance of embryogenesis before our eyes. Oh, and an update to our sand dollar larvae: they're thriving, fed the finest lab-grown algae twice a week.
We rise early and sleep early at the research station, stepping out in the mornings onto the fog-banked marina. Egrets, cormorants, and pelicans, commuters from across the bay, remind me that the sea has edges. At the start of the day, I like to walk across the bridge and back, or to visit the lookout point we found in the woods.
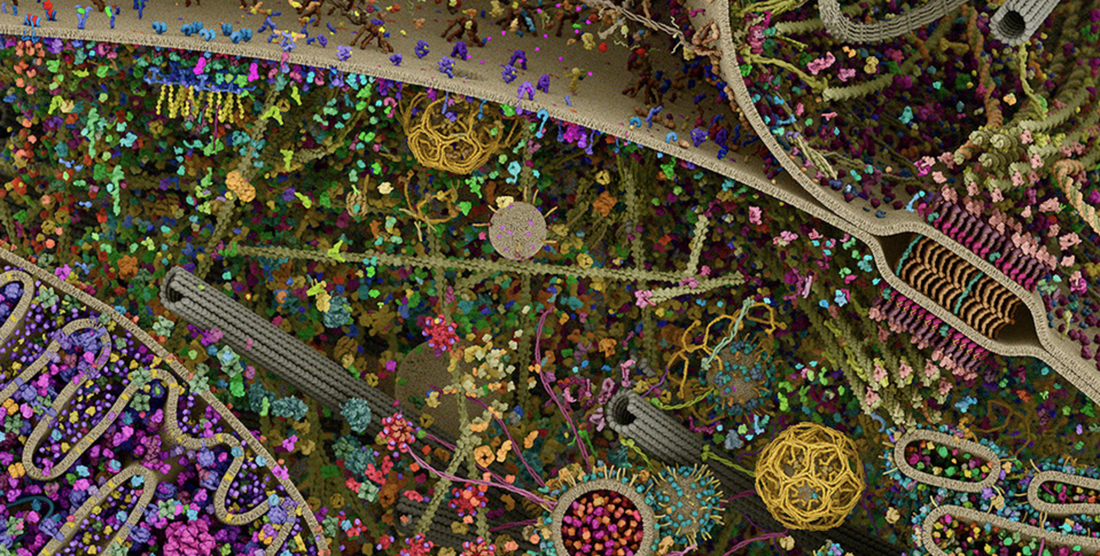
This week saw the beginnings of our projects. Chloe and I first slopped down onto the mudflats to dig up buried worms - the parents of her larval subject - and antagonize shore crabs; then we harvested sea star eggs and learned to operate the microinjector, which will be one of my main tools. For my project, I want to examine a peculiarity of cell division in sea star eggs. Cell division is a dynamic process, but it relies on a basic series of steps. Imagine that I need to bisect a circle of paper. First, I’d use a ruler and compass to orient myself with the center. Then I'd take a pen and draw a dotted guideline there. Finally, I’d fold and cut along the line. Simple as that. Animal cells, as it turns out, go about things in a similar way, using proteins. They have a folding machinery (called actin, also responsible for muscle contraction), an “inked” guideline (called Rho), a drawing hand (called Ect2), and a compass to direct it (the thready spindle structure of mitosis). But from a protein’s perspective, a cell is not a neat geometry on paper: rather a crowded, whirring industrial city. Just look at this section of the cellular landscape modeled by Evan Ingersoll & Gaël McGill. Down here, there are no straight edges, no convenient blank spaces to draw in, and nobody looking down to guide it all. Our sequence of steps, communicating only by touch, must coordinate with each other to act in synchrony across that entire city. A sea star egg cell measures about a quarter of a millimeter across - large enough for the human eye to see. It is a very big, very busy city, and sometimes the signals, the steps of division, get confused. Yet these first divisions touch off the entire undertaking of animal life. An invertebrate egg, cast out into the sea, must be especially keen to get itself divided and functional. What’s a cell to do? For the past few years, George’s lab has probed the way Ect2, Rho, and actin work together in sea star eggs. By tweaking their sensitivity, we can spark off striking ripples of activity at the cell membrane, causing Rho and actin to assemble and fall apart again in waves. It seems to be an extreme expression of a natural process, a feedback loop built into the cell. But what’s it for? Well, here’s one idea: how about an amplifier to help the machinery of division communicate across a big city?
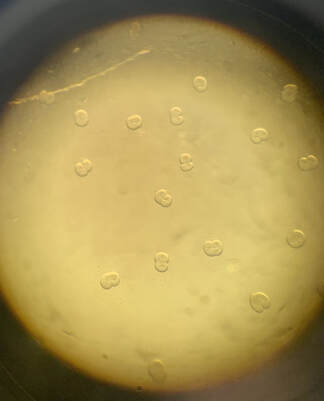
Rho waves on the surface of a sea star egg with its excitability turned up through excess Ect2 protein. This figure was published in Nature in 2015 ( Bement et al ).
Hopefully, I’ll be able to test this idea in my project this summer. We might take a sea star egg and chemically nudge Ect2 and Rho a little, then watch how the subsequent divisions go. The difficulty lies in catching it on camera… and in doing this without killing them! I wanted to practice on the fertilized eggs of the ochre star, Pisaster, a common sight in the tidepools. But after severely traumatizing nearly a dozen Pisaster specimens (gamete harvest for this species involves lopping off an arm) and finding not a single fertile male, we got fed up. We’ll be using the bat star Patiria miniata for our experiments instead. Expect to see a lot of pictures of Patiria eggs in the coming entries. Lots and lots... Greetings, readers - El here, live from the clinging fogs of Coos Bay. I’m thrilled to be part of the OIMB research community this summer. I come from Seattle, on the Salish Sea, a day’s drive north from here and a kindred ecosystem to this one. Poking around in the intertidal and the fir forest here, I realize I’m doing the same thing I’ve been doing for twenty-odd years: it was by crouching down and poking at invertebrates that I first discovered an interest in biology and art. I currently study Biology at Brown University in Providence, beside the other ocean. The Atlantic is lovely, but the chilly, smelly, alive Northwest Pacific is home to me. I’ve spent this first week getting acquainted with the fellow-creatures, human and otherwise, of OIMB. Last Saturday I moved into the dorm, alongside 10 other REU students. These fantastic people left me no reason to be nervous about meeting my coworkers, even in a building with walls that do not close all the way. We’ve gone out to explore the nearby coast and woods together, discovering secret passages, accumulating convenience store candy, and having narrow misses with toxic newts. We’ve also defeated a truly demonic puzzle. I’ll be lab partners with Chloe Goodsell; we both work under the mentorship of Dr. George von Dassow. George’s lab studies invertebrate embryos and larvae. You may witness the splendor of larval biology yourself by leaving something, maybe something expensive and clean, floating near shore for a month or so. Behold: a colorful community of life appears, seemingly from nowhere.
Cast adrift in a vast and often hostile world, an early larva must somehow form itself into an individual, and gather enough resources to maintain functionality, while surviving its habitat and its neighbors. Luckily, unlike rising college seniors, larvae are shaped by evolution to meet that challenge. I can’t wait to get to know them! George is teaching Chloe and me the tools and techniques to peer into the larval world - microscopy, organism collection, embryo culture. We’ve spent many afternoons getting close and personal with the local plankton. On Monday we also fertilized some sand dollar eggs in a dish, so in a way we’ve already created life. Two days later they’d become spaceship-like pluteus larvae, and we practiced preparing them to film. Even for a kid (me) who used to sort boxes of Cheerios for fun, manipulating individual cell clusters is a challenge. These little guys will grow through the summer, developing as our projects do. As for what the projects are? Tune back in to find out, next week! |
|
Proudly powered by Weebly




























 RSS Feed
RSS Feed